Bonding By Transfer Of Electrons Answer Key
It has to do with electron rings.
Bonding by transfer of electrons answer key. Sodium Chlorine Example B2. Complete the chart using your knowledge of atoms. A covalent bond that has an equal sharing of electrons part a is called a nonpolar covalent bond.
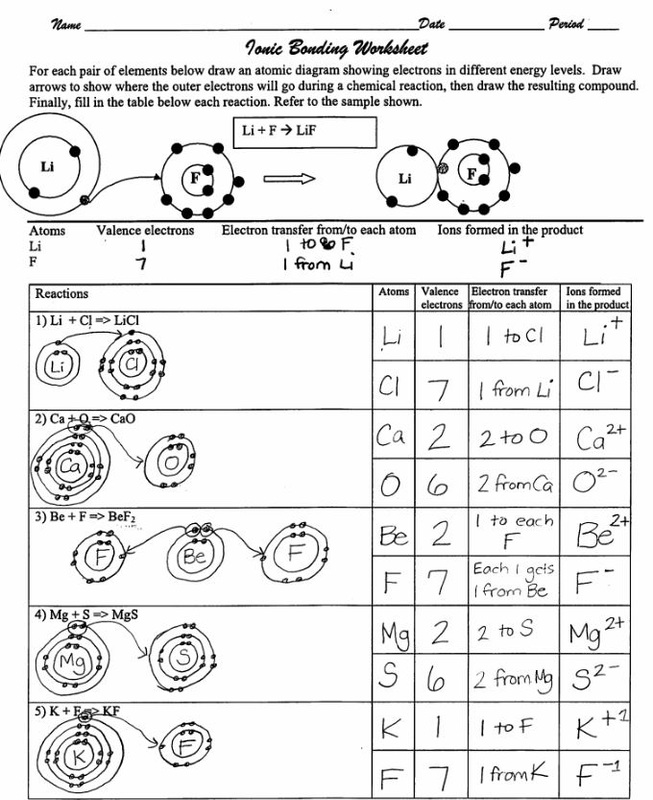
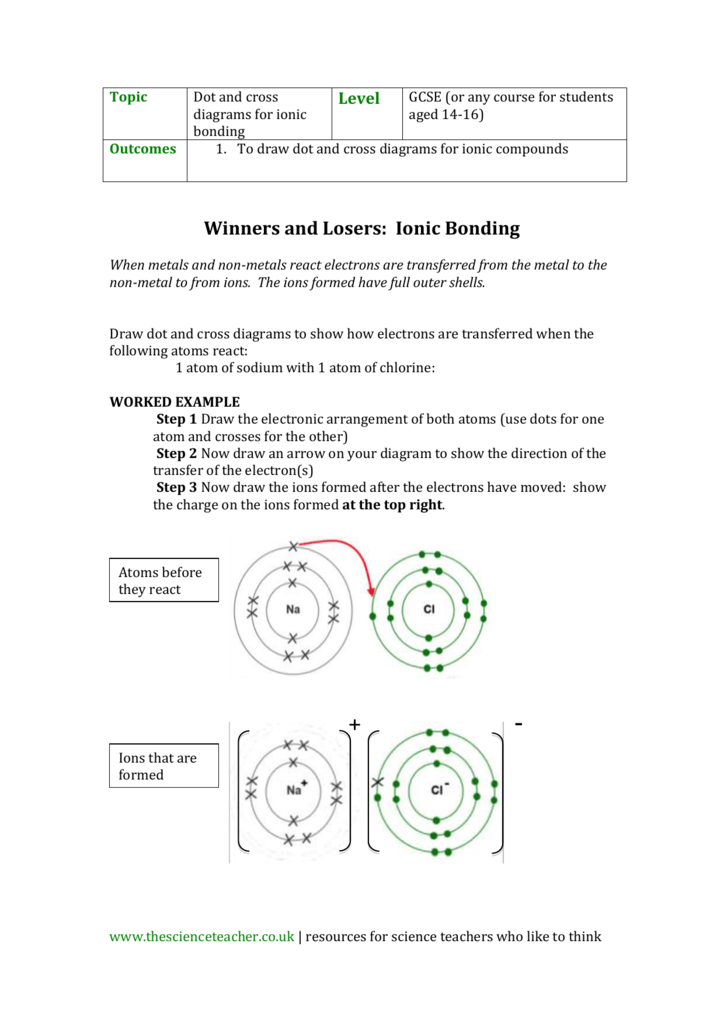
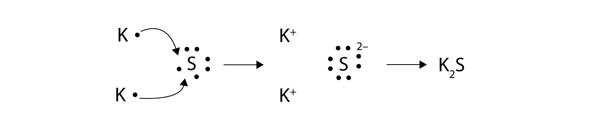
We saw this in the formation of NaCl. Balancing Chemical Equations Answer Key Gizmo Tessshebaylo It Is A Type Of Chemical Bond That Generates Two Oppositely Charged Ions. In electron transfer the number of electrons lost must equal the number of electrons gained.
Illustrate the ionic bond formed among these compounds. For each of the following compounds fill in dots to represent electrons. The transfer of electrons forms strong bonds between ions.
Ionic bonds result from the transfer of electrons from one atom to another. Spend the least money to buy the best quality products value for money. A A covalent bond is formed through the transfer of electrons from one atom to another.
The transfer of electrons results in attractive forces between molecules. This exchange results in a more stable noble gas electronic configuration for both atoms involved. Aionic because electrons are shared Bionic because electrons are transferred Ccovalent because electrons are shared Dcovalent because electrons are transferred 10Two atoms with an electronegativity difference of 04 form a bond that is ACO BKF CCaO DLiH.
Covalent bonding answer key. Solution for Activity 2. Ionic bonding is a type of chemical bond in which valence electrons are lost from one atom and gained by another.