Draw The Transfer Of Electrons Within Aluminum Chloride

The reactions for the same are given below.
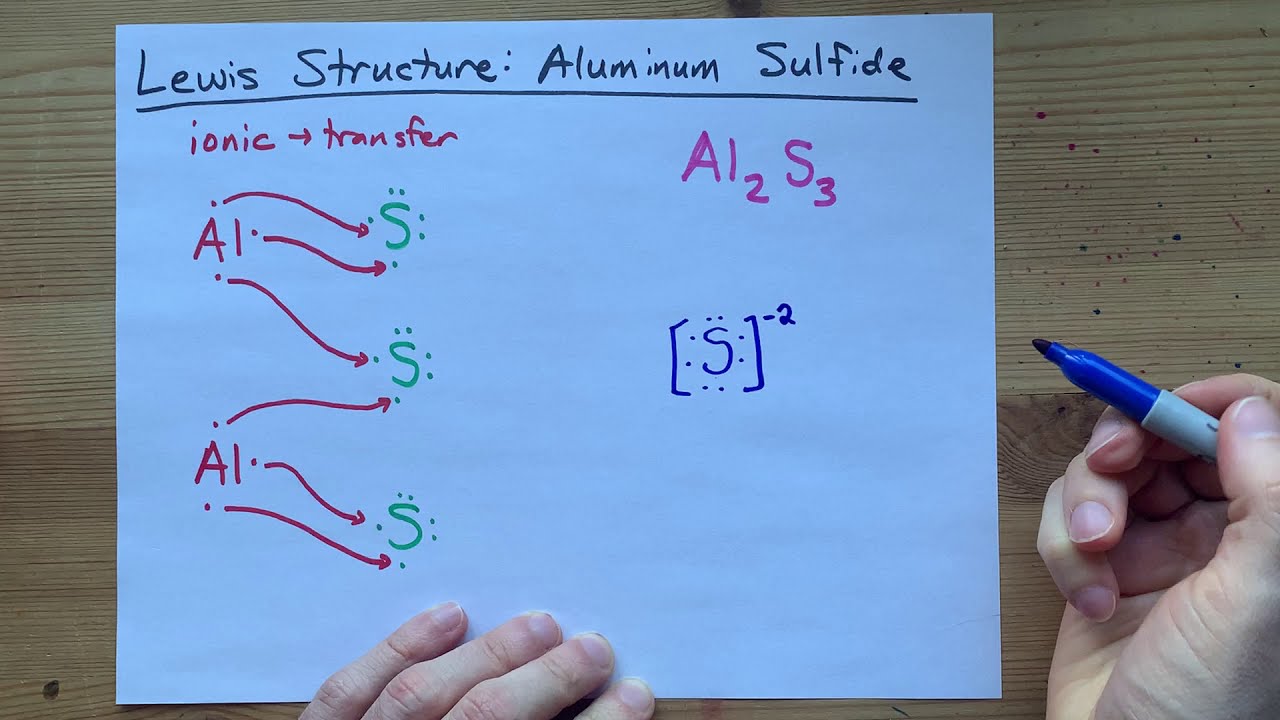
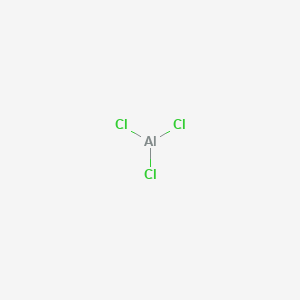
Draw the transfer of electrons within aluminum chloride. Draw the transfer of electrons within Aluminum Chloride. Its skeletal structure is given by Complete the Lewis structure and indicate the coordinate covalent bonds in the molecule. Al has three valence electrons.
Write the Chemical Formula_____ 30. Problem 120 Hard Difficulty. How are Ionic Bonds formed and what is the attractive force within an Ionic Bond.
Using Lewis Dot Structures. Ionic bonding is formed by the transfer of electrons between metal and non-metal. Write the Chemical Formula.
Draw the transfer of electrons within Aluminum Chloride. Write the Chemical Formula. 9162014 With arrows illustrate the transfer of electrons to form calcium chloride from Ca atoms and Cl atoms.
Using Lewis Dot Structures. A Cl atom needs only one more to complete its octet while Ca atoms have two electrons to lose. 222014 Aluminum and chlorine form covalent bonds not ionic bonds.
Write the Chemical Formula_____ 48. Has seven valence electrons. How are Ionic Bonds formed and what is the attractive force within an Ionic Bond.