Energy Transfer Of Ultraviolet

Chemischer Informationsdienst 1973 4 39 no-no.
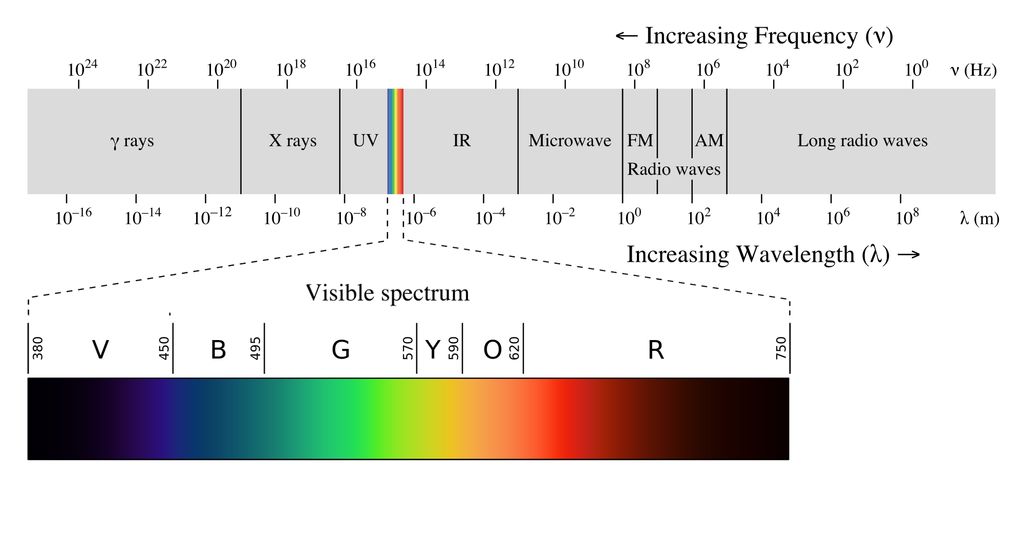
Energy transfer of ultraviolet. Quenching of singlet oxygen Guillory J. Energy of One Photon. 10 -7 centimetre or less or by the so-called trivial process of fluorescence emission from one molecule and reabsorption by one at any distance.
The development of sunscreens with high sun protection factor SPF values but low filter concentrations is the ultimate goal. It can be expected that during this process free radicals are produced. 312021 In general the resonant-type energy transfer occurs through an exchange interaction or electric multipolar interaction.
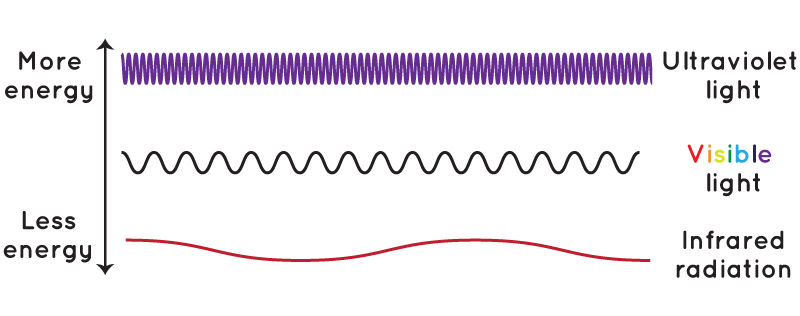
As frequency increases into the ultraviolet photons now carry enough energy about three electron volts or more to excite certain doubly bonded molecules into permanent chemical rearrangement. 1012011 Consequently it can be assumed that an energy transfer from the UV filter to the ingredient Tetradodium EDTA takes place during UV irradiation. The crystal structure luminescence and energy transfer of Ce 5 Si 3 O 12 NTb 3 were investigated in detail.
The Planks constant is the parameter which is. 980808 nm dual-wavelength responsive. This design ensures the entire surrounding of emitters eg Tm 3 Er 3 and Gd 3 by the sensitizers which is able to increase the localized excitation energy density and further accelerate the population of intermediate energy levels as well as the higher-lying UV emitting level by sequential energy transfer upconversion processes.
The transfer of UV photon energy into fluorescent light represents a new approach to increase the efficiency of sunscreens and could form the basis for a new generation of. Ultraviolet visible spectroscopy λ 200 - 800 nm studies the changes in electronic energy levels within the molecule arising due to transfer of electrons from π- or non-bonding orbitals. The energy transfer from Eu 2 to Mn 2 has been demonstrated to be a resonant type via a dipole-quadrupole mechanism.
Interestingly Ce 5 Si 3 O 12 N has self-luminescent properties and its excitation falls in the near-ultraviolet region and emits a broad band cyan light which belongs to the 5d4f transition of Ce 3 ions. The energy of one photon can be calculated as the product of the frequency of the photon and the Planks constant. For complete understanding a brief introduction to electron transfer reactions and Marcus-Hush theory is necessary.