How To Show Transfer Of Electrons

Mg 28 Mg2 2e-.
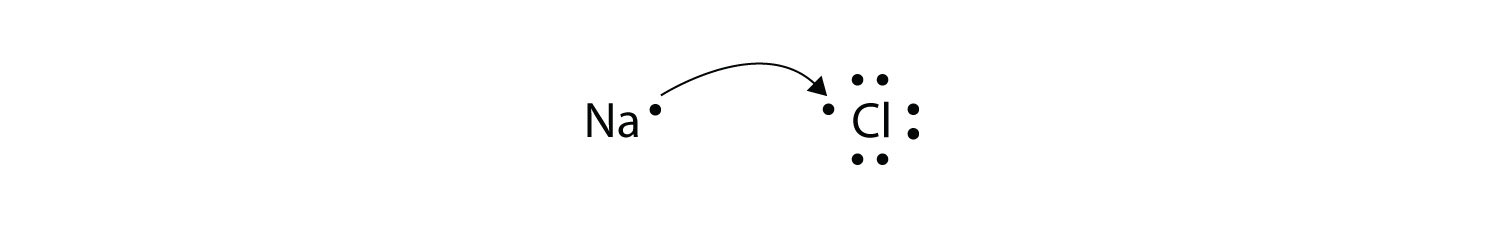
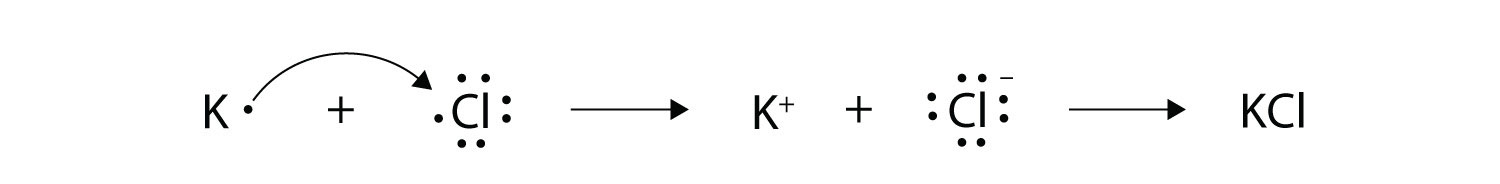
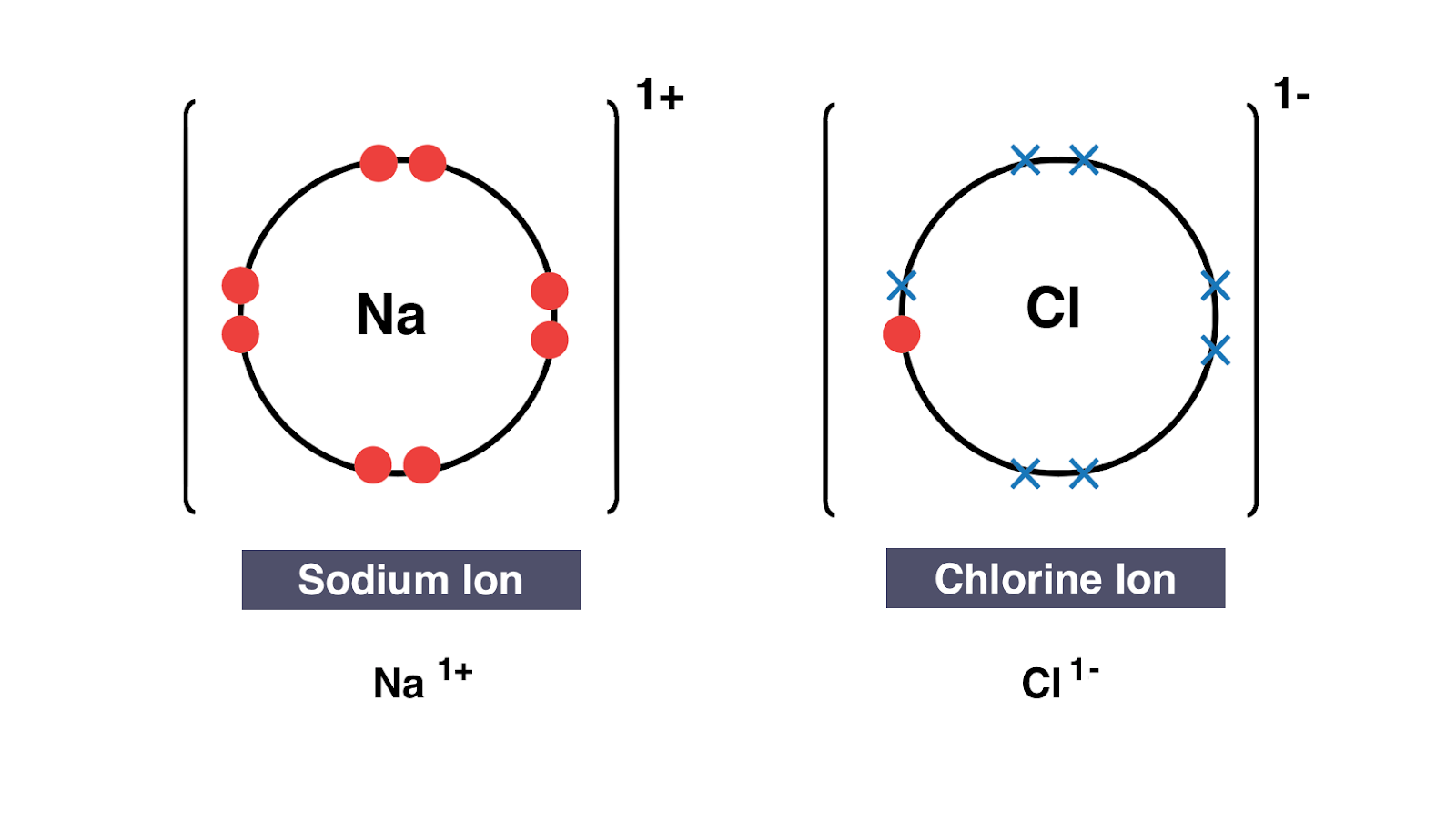
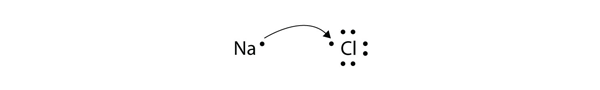
How to show transfer of electrons. 288 hence valency -1 Formation of MgCl2. Ionic bonds involve a cation and an anion. Another atom typically a non-metal is able to acquire the electrons to become a negative ion or anion.
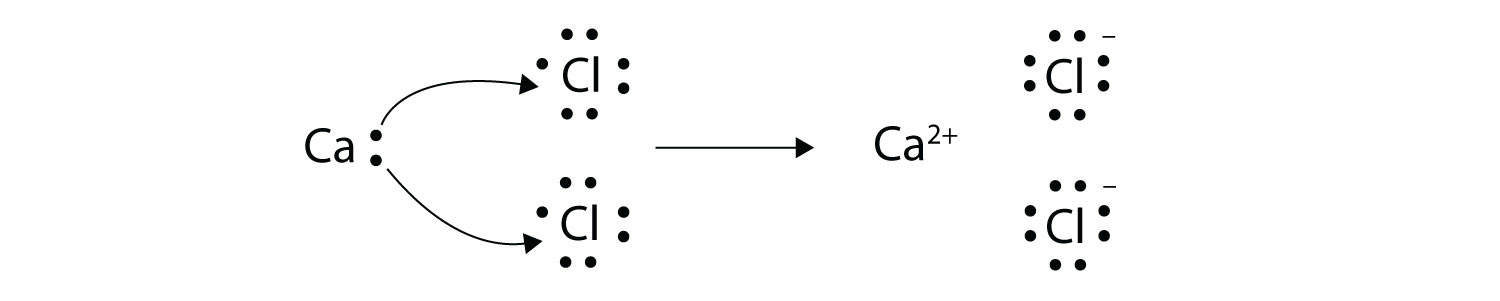
With arrows illustrate the transfer of electrons to form calcium chloride from Ca atoms and Cl atoms. So in our example here were given Cu2 plus Br- yields CU plus BrO3-. Shows the movement of an electron pair.
Because the oxidation numbers changed an oxidationreduction reaction is defined as one in which electrons are transferred between atoms. The bond is formed when an atom typically a metal loses an electron or electrons and becomes a positive ion or cation. A Cl atom needs only one more to complete its octet while Ca atoms have two electrons to lose.
Conduction occurs when there is direct contact between materials that differ in their ability to give up or accept electrons. Show the Formation of Na2o by the Transfer of Electrons Between the Combining Atoms. A Cl atom needs only one more to complete its octet while Ca atoms have two electrons to lose.
The first essential rule to keep in mind is the following. 9162014 With arrows illustrate the transfer of electrons to form calcium chloride from Ca atoms and Cl atoms. So Ill actually show you two ways.
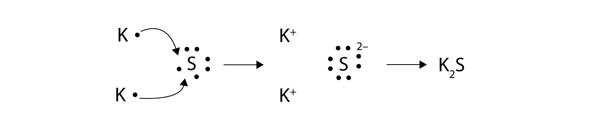
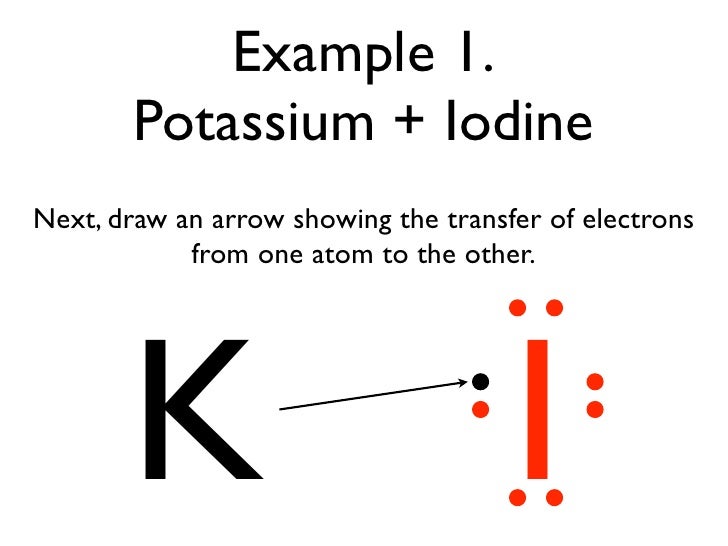
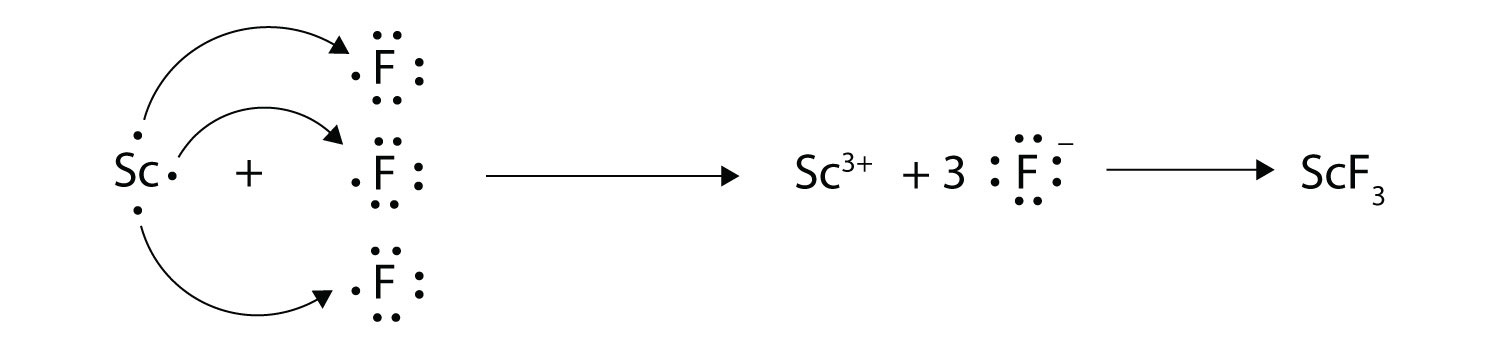
1042017 Use Lewis symbols to show electron transfer between the following atoms to form cations and anions. Chemical bonding and molecular structure. Electronic configuration 287 Stable form.