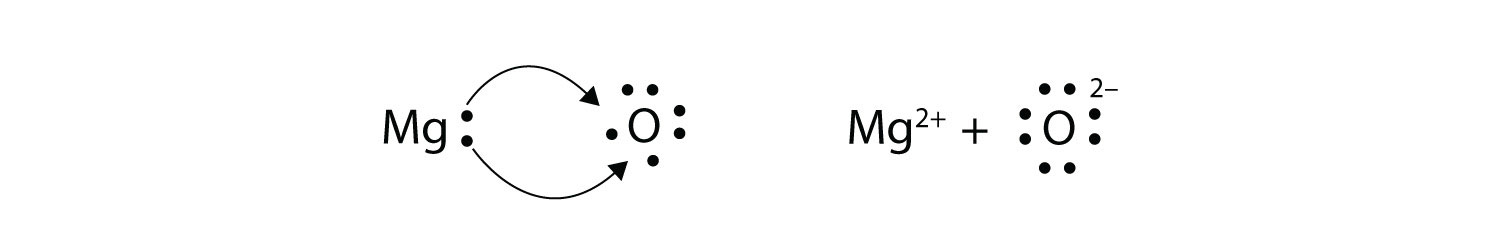Ions Transfer Of Electrons Lab 6

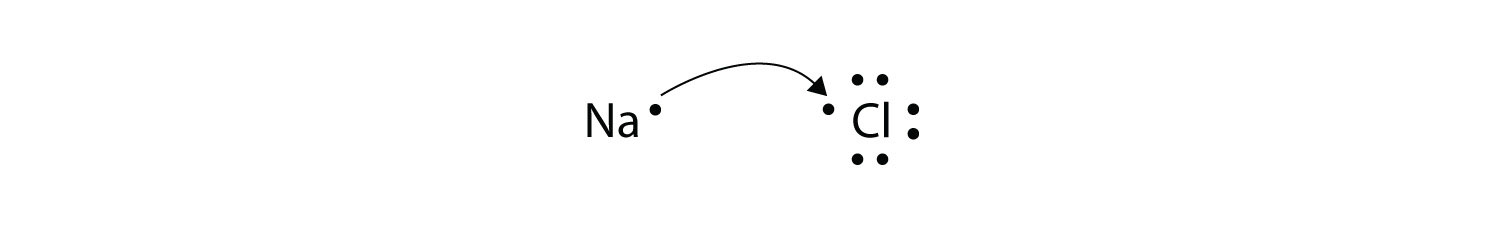
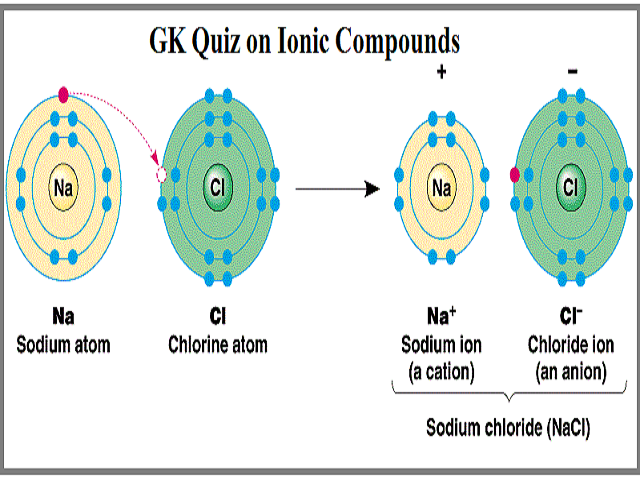
A A sodium atom Na has equal numbers of protons and electrons 11 and is uncharged.
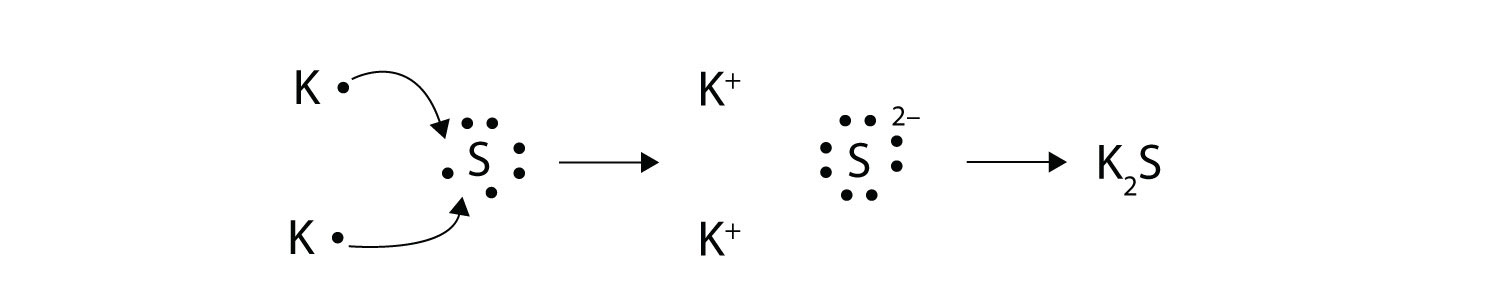
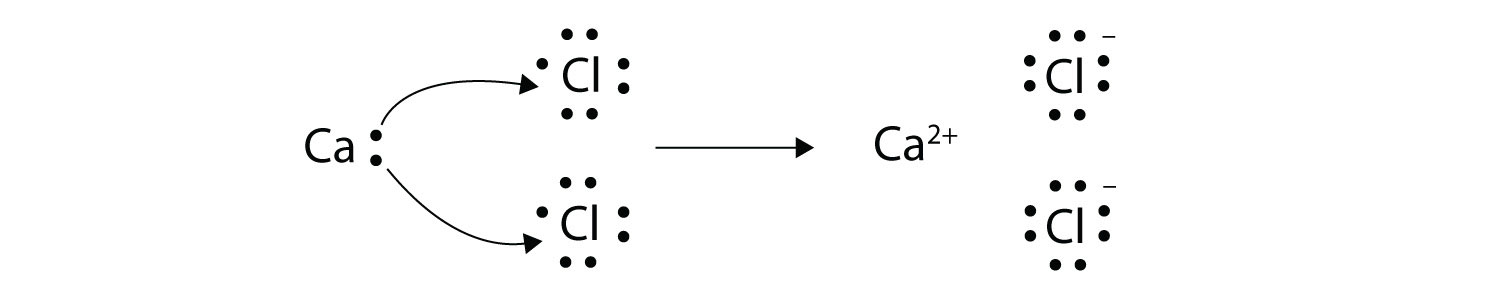
Ions transfer of electrons lab 6. Chapter 6 61 Ions. We saw this in the formation of NaCl. Section 61 - Ions.
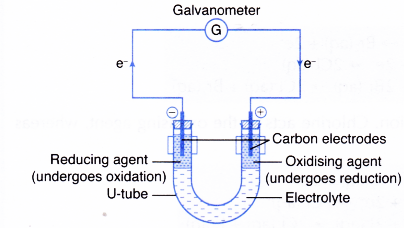
The loss of electrons is called oxidation. Transfer of Electrons - Study Check 61. Atoms will lose gain or share valence electrons to acquire an OCTET.
The ions produced are oppositely charged and are attracted to one another due to electrostatic forces. Of 8 valence electrons. Section 61 - Ions.
The gain of electrons is called reduction. Transfer of Electrons Element Atemi 1. Lose electrons to non-metal atoms with 5-7 electrons missing.
Formed when atoms lose gain or share valence electrons to acquire an octet of 8 valence electrons. The process by which an atom or a molecule acquires a negative or positive charge by gaining or losing electrons to form ions often in conjunction with other chemical changes. An Introduction to General Organic and Biological Chemistry 12th Edition answers to Chapter 6 - Section 61 - Ions.
Electron Configuration of Atom 11 122. The resulting Na and F ions are electrically attracted to each other. Name of los of lon I.