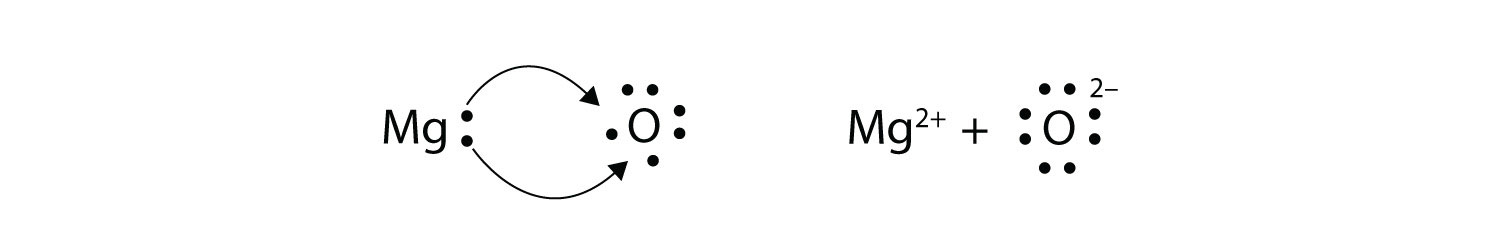Ions Transfer Of Electrons Lab 9
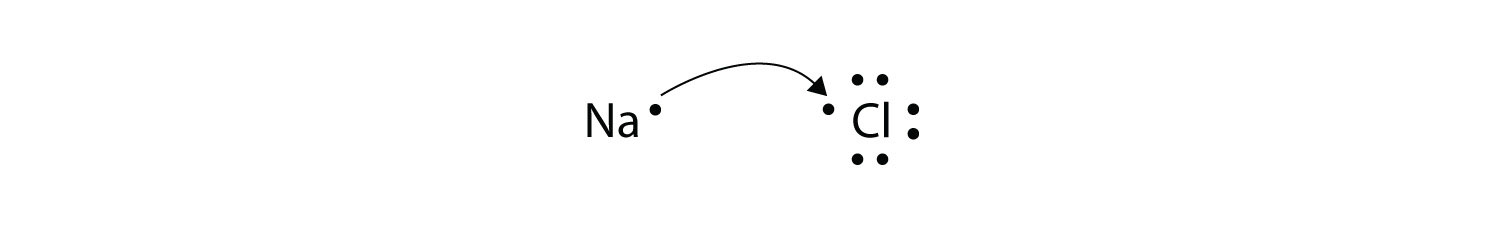
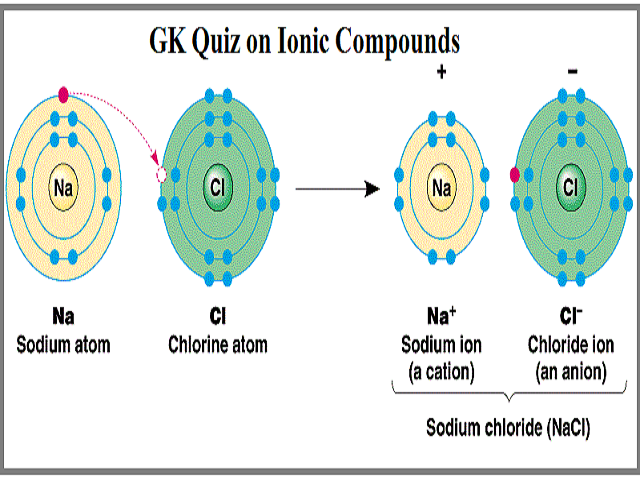
Ionic bond electron from Na is transferred to Cl this causes a charge imbalance in each atom.
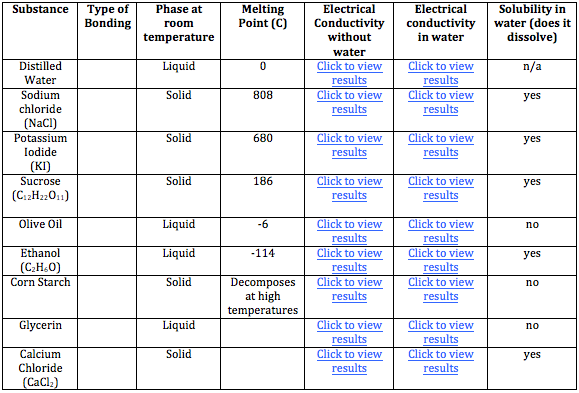
Ions transfer of electrons lab 9. NONPOLAR COVALENT BONDS H2 or Cl2 2. Demonstrate electron transfer between atoms to form ionic bonds. Example of ion charges and groups.
Covalent bonds- Two atoms share one or more pairs of outer-shell electrons. In the last shell of an atom. For example when a strip of solid Zinc metal is placed in a solution of copper ions the zinc metal begins to dissolve and the copper metal begins to deposit onto the zinc metal according to the following chemical reaction.
Ionic Bonding and Electron Transfer Ions are atoms or molecules bearing an electrical charge. Im text Arial BIUA 5 23 LAB REPORT SHEET Compounds and Their Bonds 9 A. Transfer of Electrons Element Atomi c Num-ber 1.
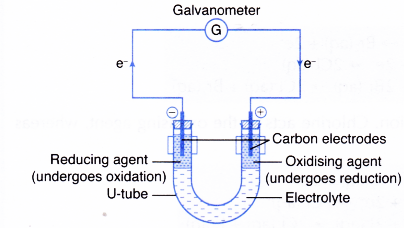
How specifically are positive and negative ions formed. The gain of electrons is called reduction. 9-1 Experiment 9 Electrochemistry I Galvanic Cell Introduction.
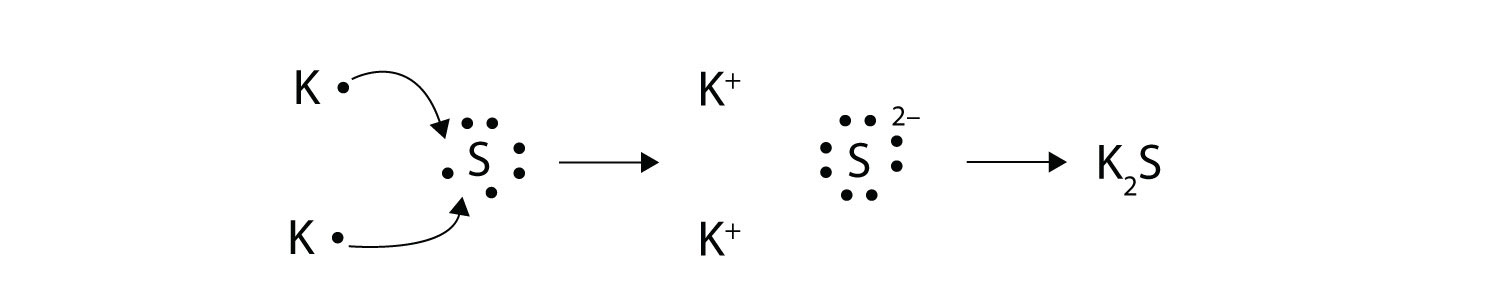
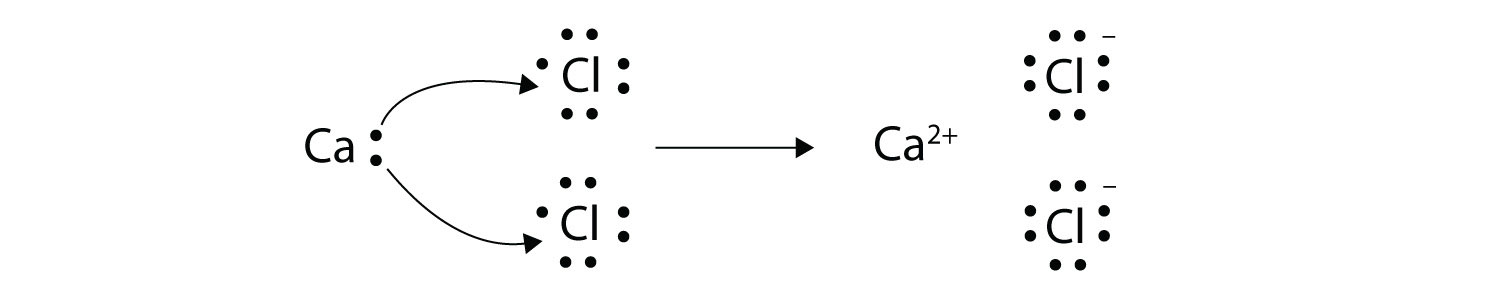
Ions are formed by the transfer of electrons. Many of the ions that form have eight electrons in their valence shell. When a stable atom gains or loses an electron it becomes an ion.
Ionic bonds are caused by electrons transferring from one atom to another. So when an electron is lost on group 1A 1 electron is lost making a positively charged ion. In electron transfer the number of electrons lost must equal the number of electrons gained.