Show The Transfer Of Electrons Kf

A Lewis electron dot diagram or electron dot.
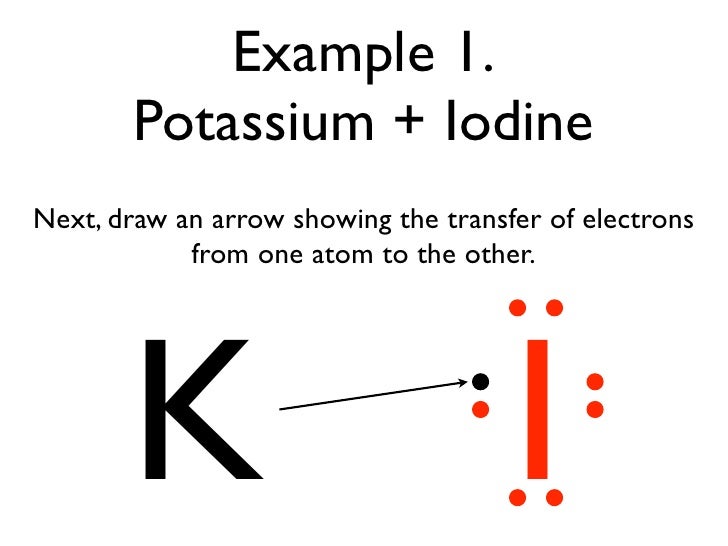
Show the transfer of electrons kf. We saw this in the formation of NaCl. Ionic bonds are caused by electrons transferring from one atom to another. A K and S b C a and O c A l and N.
Complete the questions below showing the transfer of electrons between metal and nonmetal the Lewis structure of the resulting compound the formula unit and the name of the compound remember the anion name ends with ide. B Ca and O. C Al and N.
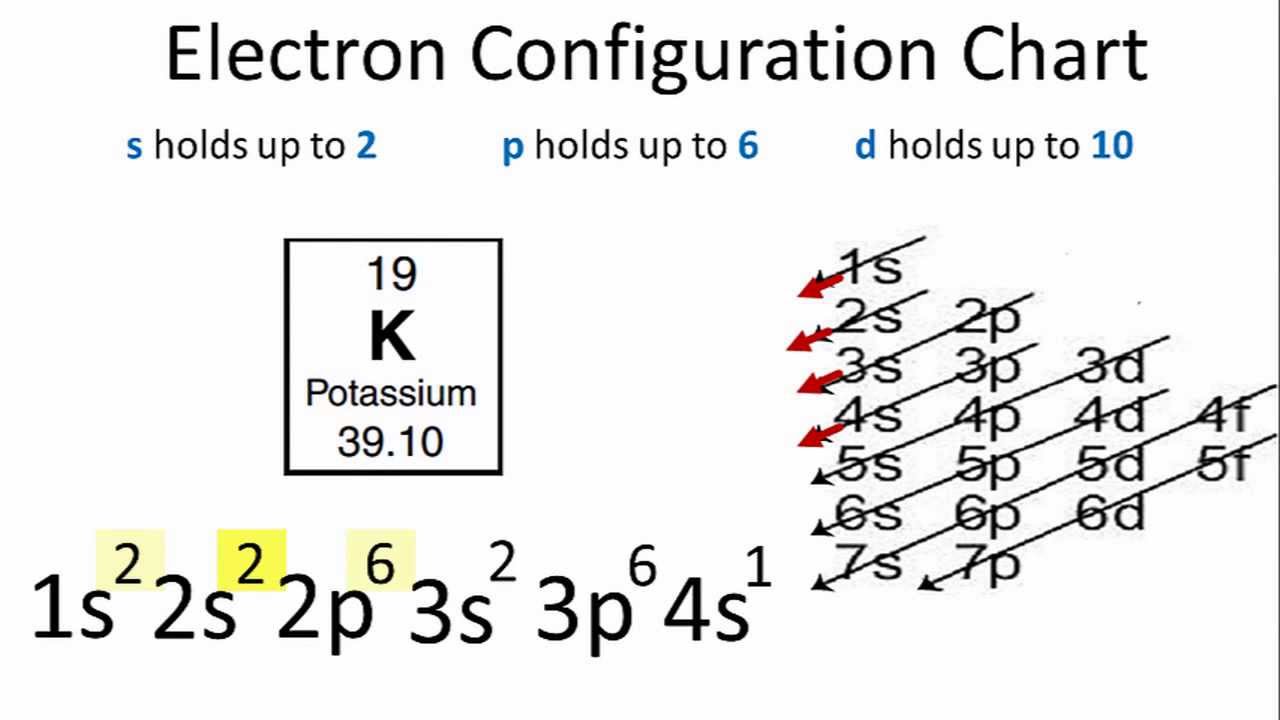
We can use electron configurations to illustrate the electron transfer process between sodium atoms and chlorine atoms. These ions have a negative charge. In almost all cases chemical bonds are formed by interactions of valence electrons in atoms.
In electron transfer the number of electrons lost must equal the number of electrons gained. Sodium and oxygen 6. During chemical changes an oxygen atom gains 2 electrons in the sec-ond energy level from another atom.
The electronic configurations of Ca and O are as follows. In electron transfer the number of electrons lost must equal the number of electrons gained. 6122010 Hence the electron transfer can be shown as.
Al gives 3 and 3Br each accept 1-. The equilibrium constant K for the cell is given by where F is the Faraday constant R is the gas constant and T is the temperature in kelvins. Na gives 1 but must be 2 atoms because O accepts 2-.