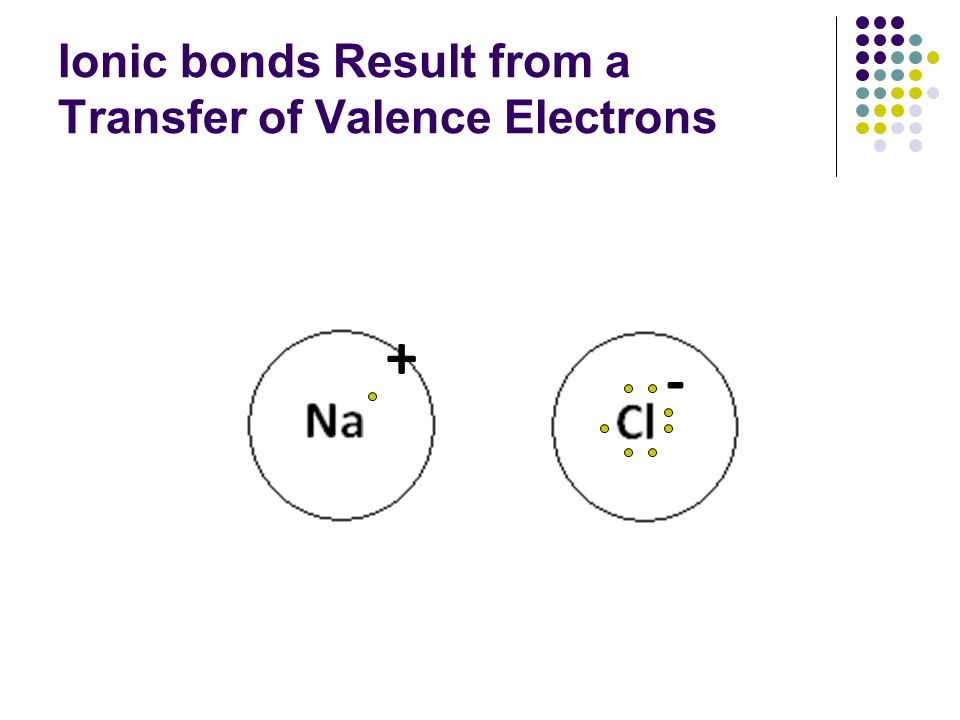
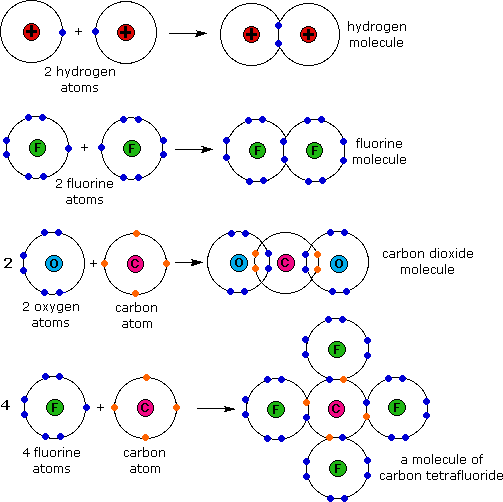
The Transfer Of Valence Electrons Results

By considering the recorded image as a CTF-degraded true object describing the CTF allows the.
The transfer of valence electrons results. This reaction is mediated by a nuclear coordinate q. Oxidation is a process in which an atom or a group of atoms taking part in chemical reaction loses one or more electrons. According to experimental results and density functional theory calculation there were van der Waals interaction and a part of the strong attraction between the interface of PS and Mn 3 O 4 C which could be enhanced by inner Mn 3 O 4 and thus promoted the electron transfer between PS and carbon shell and the defective edges of the carbon.
We may note that one-electron transfer gives rise to products corresponding to the transfer. Electron transfer results in products with new interest-ing properties. The contrast transfer function mathematically describes how aberrations in a transmission electron microscope modify the image of a sample.
A cation a positive ion forms when a neutral atom loses one or more electrons from its valence shell and an anion a negative ion forms when a neutral atom gains one or more electrons in its valence. The process of efficiently and controllably moving electrons around is one of the primary regulation mechanisms in biology. Our results confirm the ET mechanism as hole transfer HT through residues Ala194 Ala193 Gly192 and Trp191 of CcP.
Electron transfer processes has become possible only when new instrumental research techniques particularly ESR NMR and microwave spectroscopy of chemically polarised nuclei have been introduced into chemical practice. The changes in oxidation states of the donor and acceptor centers result in a change in their equilibrium nuclear configurations. As shown by experiments in the presence of acetate this rate-limiting step cannot be overcome simply by applying a stronger potential.
Compare Cophen3 23 and CoNH 36. 4282020 Interestingly the electrons released from the NH 4 oxidation were transferred to the extracellular electron acceptor via a pathway that is analog to the ones present in. Oneidensis thereby enhancing intracellular electron flux and the EET rate.
These results reflect the findings of Marcus theory of electron transfer. Electron transfer or the act of moving an electron from one place to another is amongst the simplest of chemical processes yet certainly one of the most critical. The process of transference of electrons is described as redox process.