Transfer Electrons Heat

Special effort then follows to extract the commonalities among the.
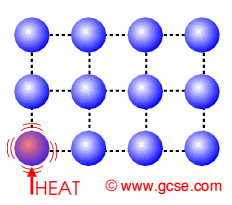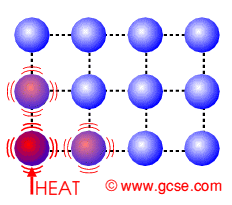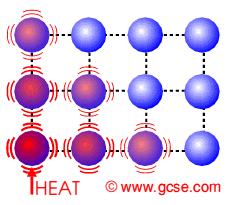
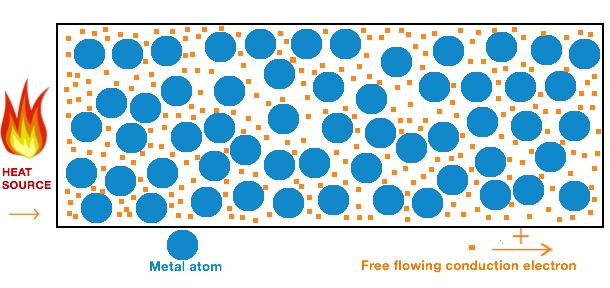
Transfer electrons heat. How do metals conduct heat. These vibrations pass from atom to atom transferring heat energy as they do so. Specific heat just like conductivity depends on the molecular structure of the solid and that depends on the atomic number ie.
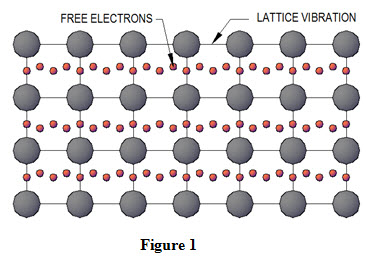
They have the same number of molecules. Electrons can also carry heat which is the reason metals are generally very good conductors of heat. Heat Transport by Phonons and Electrons 3.
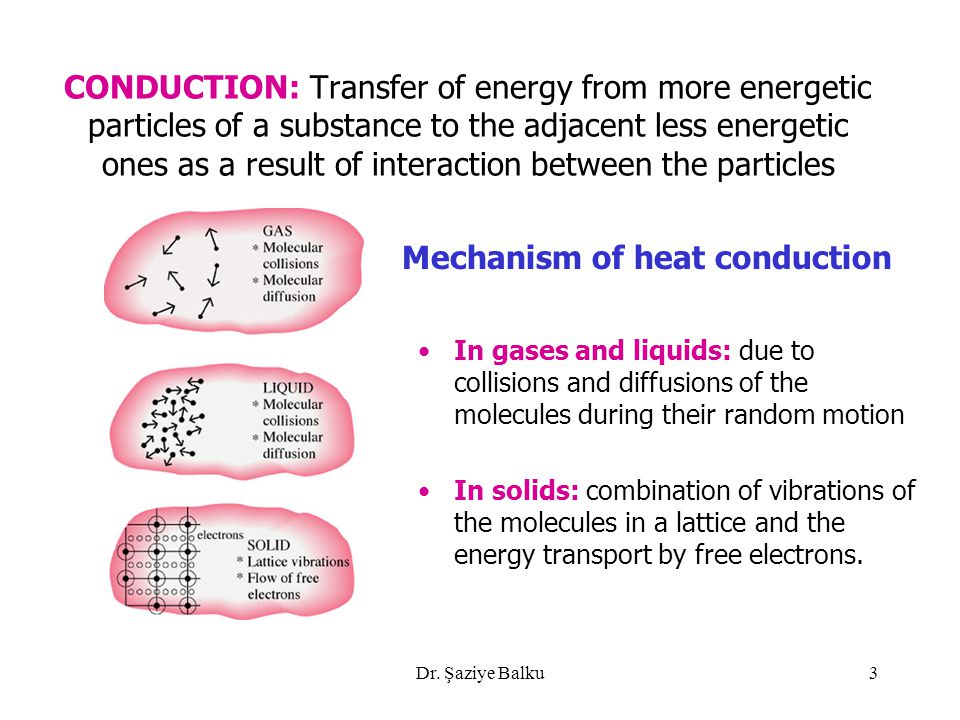
Heat transfer in liquids and gases is essentially due to_____ a Conduction b Convection c Radiation d Conduction and convection put together. Insulators do not have free electrons and so they do not conduct heat as well as metals. There are three modes of heat transfer.
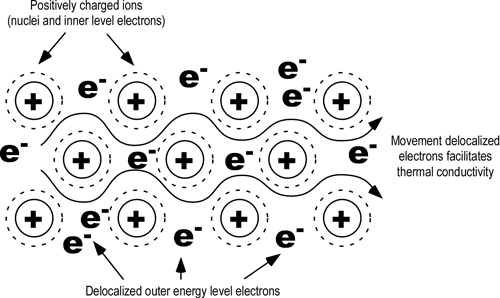
The electrons transfer the heat energy by colliding with other atoms and electrons in the metal see also the theory of electrical resistance. Rather heat is transferred internally by vibrations of atoms and molecules. Conduction is a process of heat transfer from.
The electrons in piece of metal can leave their atoms and move about in the metal as free electrons. Thermal Conduction of metals will include molecular collisions conduction electrons for metals in gas state and lattice vibrations conducting electrons for metals in solid state. It can occur in solids liquids and gases.
In the above picture the. In another ward conduction is the transfer of energy from the more energetic to less energetic particles of a substance due to interaction between the particles. An extreme close-up on heat transfer.