Transfer Of Electrons Between Alkali Metals And Halogens
The only way to achieve stability is to have a full outer valence shell If the periodic table was cylinder think that the further you are from noble hasses.
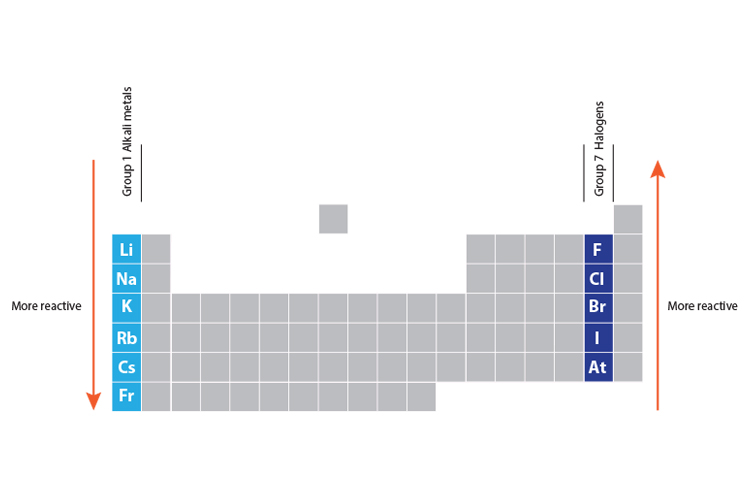
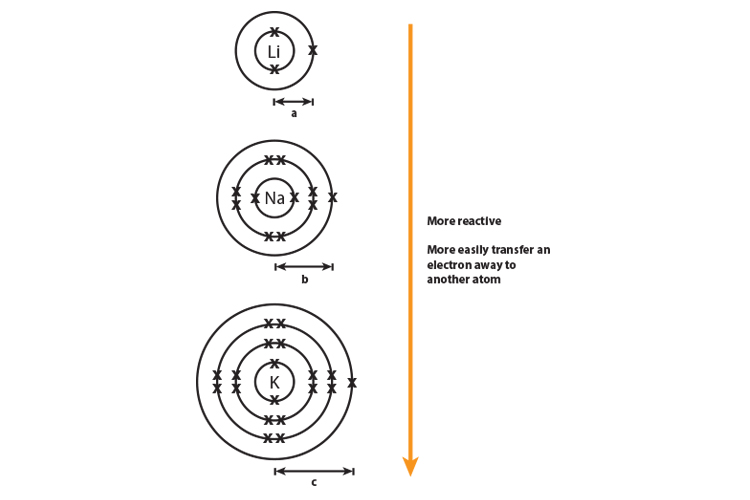
Transfer of electrons between alkali metals and halogens. Each element from halogens replaces the elements below it in their salt solution. Alkali metals and alkaline earth metals. Alkali metals have 1 electron in their valence shell so they tend to lose 1 electron to fill their previous octet.
Each alkali metal has a single electron. See full answer below. Removes air traces in vacuum tubes.
As such the number of valence electrons is A. 4232019 Alkali metals belong to the s-block elements occupying the leftmost side of the periodic tableAlkali metals readily lose electrons making them count among the most reactive elements on earth. 11282020 Halogens react with metals forming salts so they are called halogens which means Salts formations.
In this article we will explain the electronic configurations ionization enthalpy hydration enthalpy and atomic ionic radii and other physical and chemical properties of the group one alkali metals. 952020 Figure 4 1 Periodic Table Of Elements Identifying Alkali Metals Alkaline Earth Halogens Le Gases Metalloids Nonmetals Reative And Transition Diagram Quizlet. The Group 1 elements in the periodic table are known as the alkali metals.
Halogens have 7 electrons in their valence shell so they tend to gain 1 electron to fill their octet. These elements tend to have high reactivity with their ability to remove electrons from other species. The reactivity of Group 7 elements decreases down the group.
For a group number less than 5 the group number is simply the number of valence electrons. Alkaline earth or alkali metals the halogens tend to gain electrons to form anions X 1-. Nature of the Metal.