Transfer Of Electrons Between Atoms Results In The Formation Of

5212020 The transfer and sharing of electrons among atoms govern the chemistry of the elements.
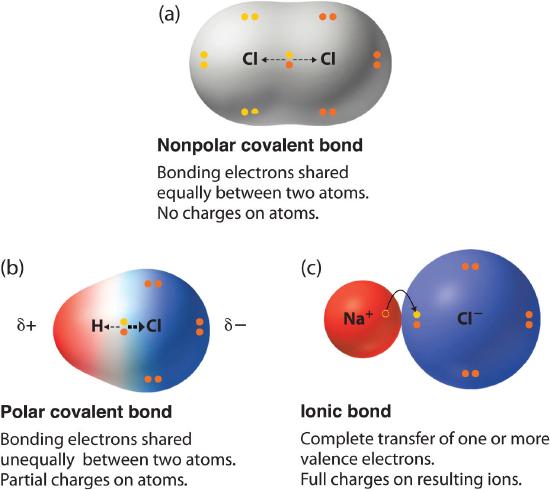
Transfer of electrons between atoms results in the formation of. Cl287 e- Cl-When 2 free chlorine atoms accept the electrons given by single magnesium ion the compound MgCl 2 is formed. Click hereto get an answer to your question Show on a diagram the transfer of electrons between the atoms in the formation of MgO. Ionic bonding is a type of chemical bond in which valence electrons are lost from one atom and gained by another.
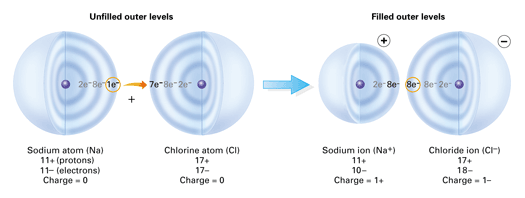
In electron transfer the number of electrons lost must equal the number of electrons gained. In the process of either losing or gaining negatively charged electrons the reacting atoms form ions. Chlorine on the other hand has 7 electrons in its valence shell and needs one more in order to complete its octet.
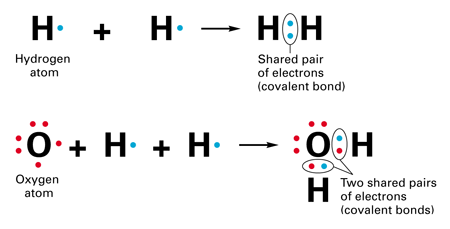
For example consider carbon tetrachloride. It means that the. This sharing of electrons results in bond formation between the two atoms.
A similar process occurs between Mg atoms and O. In ionic bonding electrons are completely transferred from one atom to another. Ionic bonds are caused by electrons transferring from one atom to another.
A similar process occurs between Mg atoms and O. Show the Formation of Na2o by the Transfer of Electrons Between the Combining Atoms. 10192018 Ionic bonding results from the electrostatic attraction of oppositely charged ions that are typically produced by the transfer of electrons between metallic and nonmetallic atoms.
We saw this in the formation of NaCl. This results in the formation of ions - positive ions from the metal atoms and negative ions from the non-metal atoms. The atom with the higher electronegativity will pull more electrons from an atom with lower elctronegativity.