Transfer Of Electrons Between Magnesium And Chlorine

2132011 Magnesium will be having two extra electrons in the outer shell hanging waiting to be transferred.
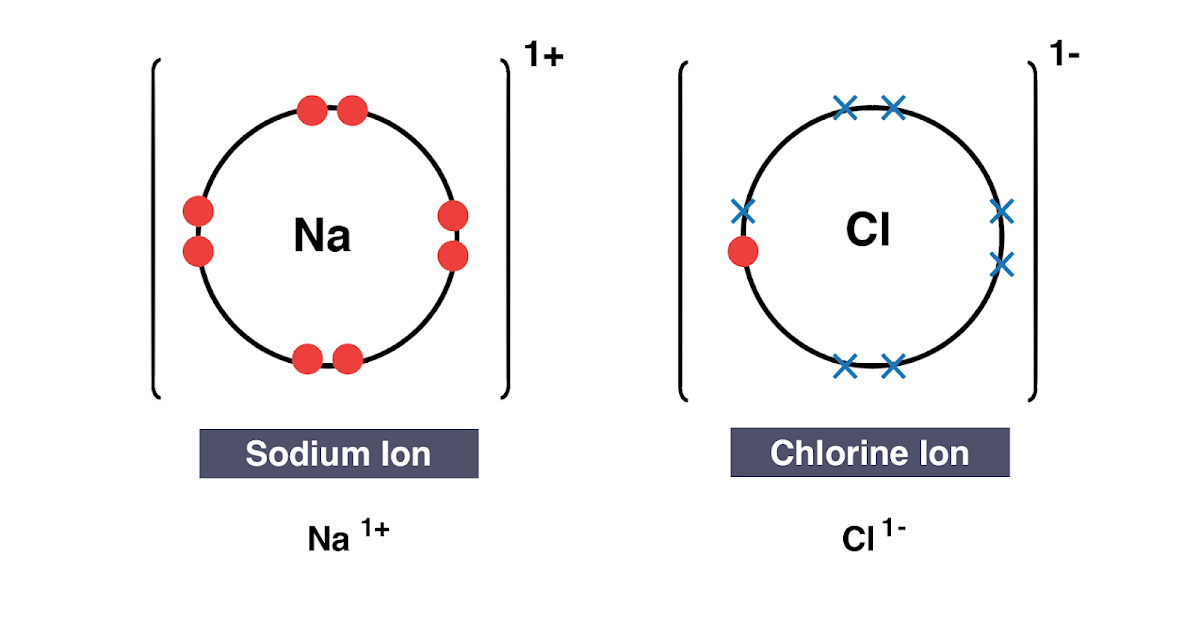
Transfer of electrons between magnesium and chlorine. 9162014 Ionic bonds are caused by electrons transferring from one atom to another. Atoms with relatively similar electronegativities share electrons between them and are connected by covalent bonds. CBSE CBSE Class 10.
Show the formation of calcium chloride by transfer of electrons. Magnesium atom loses two electrons to form magnesium cation where as each chlorine atom accepts one electron to form chloride ion anion. Show the formation of magnesium chloride by the transfer of electrons Acids Bases and Salts-Science-Class-10.
Click to see full answer. A similar process occurs between Mg atoms and O atoms except in this case two electrons are transferred. Advertisement Remove all ads.
Show the Formation of Magnesium Chloride from Magnesium and Chlorine by the Transfer of Electrons. Show the formation of magnesium chloride from magnesium and chlorine by the transfer of electrons. D Ionic Bond is the type of chemical bonds are present in the magnesium chloride formed.
The atomic number of magnesium is 12 and of chlorine is 17. 17 and calcium At No. 10262020 Formation of chlorine ion by accepting an electron Cl 287 e- Cl- When 2 free chlorine atoms accept the electrons given by single magnesium ion the compound MgCl2 is formed.
A similar process occurs between Mg atoms and O atoms except in this case two electrons are transferred. The oppositely charged of the magnesium and chloride ions attract each other and ionic bonds are formed. Show the formation of magnesium chloride by the transfer of I electrons.