Transfer Of Electrons Between Sodium And Oxygen

Please log in or register to add a comment.
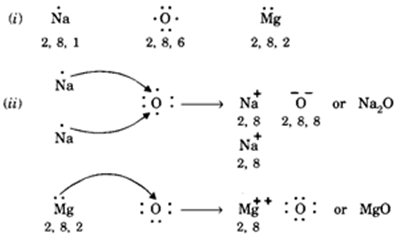
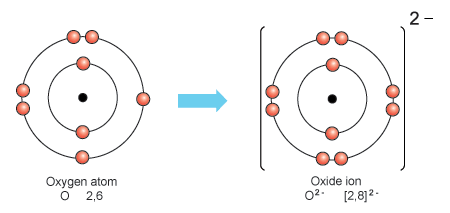
Transfer of electrons between sodium and oxygen. I Write the electron-dot structures for sodium oxygen and magnesium. Sodium ions and oxide ion being oppositely charged attract each other and are held together by strong electrostatic forces of attraction to form the ionic sodium oxide compound 2Na O 2- or Na 2 0. During the bond formation between sodium and oxygen two sodium atoms loses one electron each to oxygen atom 2N a2e 2N a O 2e O2 2N a O2 N a2.
712008 One oxygen atom will gain both of these electrons. The chemical species from which the electron is removed is said to have. Na2O contains Na and O2 ions.
One O2- ion and two Na ions will form. Two sodium atoms will each gain one of these electrons. Two Na ions and one O2- ion will form.
Electronic configuration of sodium -. However when we dissolve the ionic solid in water or melt it the crystal structure is broken down and ions become free to move and conduct electricity. Iii They conduct electricity in molten state as well as in aqueous solution.
10122016 This Lewis dot structure shows the shared. It is two electrons short of octet configuration thus it will gain two electrons. In the aluminumoxygen example the aluminum was oxidized and the oxygen was reduced because every electron transfer reaction involves simultaneous oxidation and reduction.
In electron transfer the number of electrons lost must equal the number of electrons gained. In this process both the atoms sodium and oxygen obtain the stable electronic configuration of the noble gas neon. I They are solids having high melting point.