Transfer Of Electrons Called
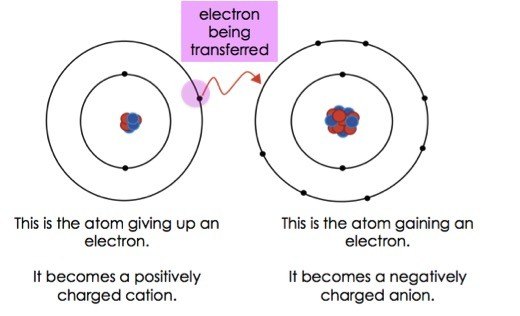
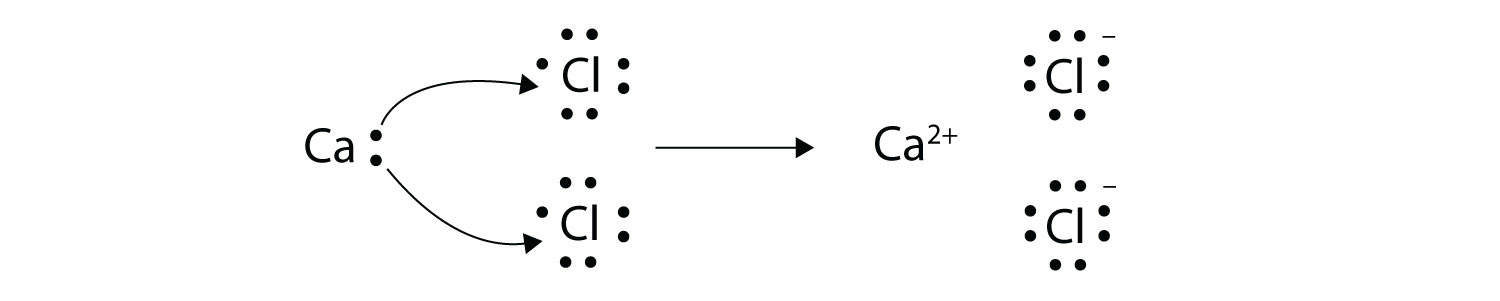
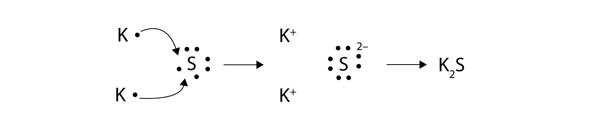
1272021 The attraction between oppositely charged ions is called an ionic bond and it is one of the main types of chemical bonds in chemistry.
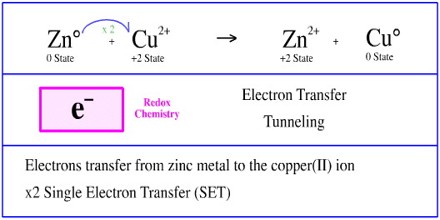
Transfer of electrons called. In the example each oxygen atom has gained two electrons and each aluminum has lost three electrons. In electron transfer the number of electrons lost must equal the number of electrons gained. The transfer of electrons from one object to another by direct contact.
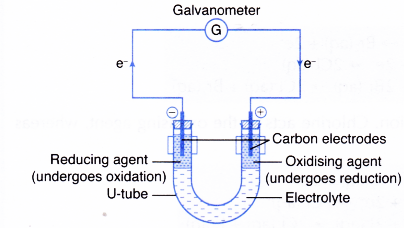
This process is called redox process. This process creates a proton gradient also known as the electrochemical gradient. 9162014 The attraction between oppositely charged ions is called an ionic bond and it is one of the main types of chemical bonds in chemistry.
Now let us understand the definition of oxidation and reduction according to electron transfer. This is known as electron-transfer ET. 2Covalent bond- Some electrons from each atom are shared by all the atoms such that those electrons complete the octet of each contributing atom.
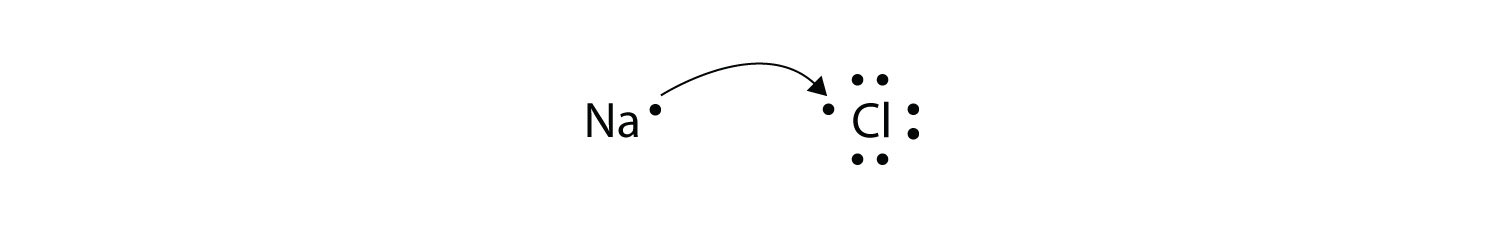
We saw this in the formation of NaCl. Ionic bonds are caused by electrons transferring from one atom to another. Oxidation - reduction reaction Single Replacement Reactions is antoher type of ________ reaction.
The attraction between oppositely charged ions is called an ionic bond and it is one of the main types of chemical bonds in chemistry. A bond formed by the transfer of electrons between atoms of the elements is called- Click here for Free SSC Mock test by topic A bond formed by the transfer of electrons between atoms of the elements is called- 1 ionic bond. When such a transfer occurs all the valence electrons on the more electropositive element from one of the first three groups on the left in the periodic table are removed to expose the core of the atom.
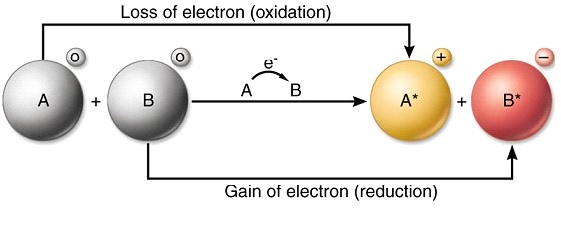
A A sodium atom Na has equal numbers of protons and electrons 11 and is uncharged. Because the oxidation numbers changed an oxidationreduction reaction is defined as one in which electrons are transferred between atoms. The transfer and sharing of electrons among atoms govern the chemistry of the elements.