Transfer Of Electrons Can Be The Driving Force For A Reaction

Transfer of electrons formation of a gas formation of a solid or dissolving of a salt.
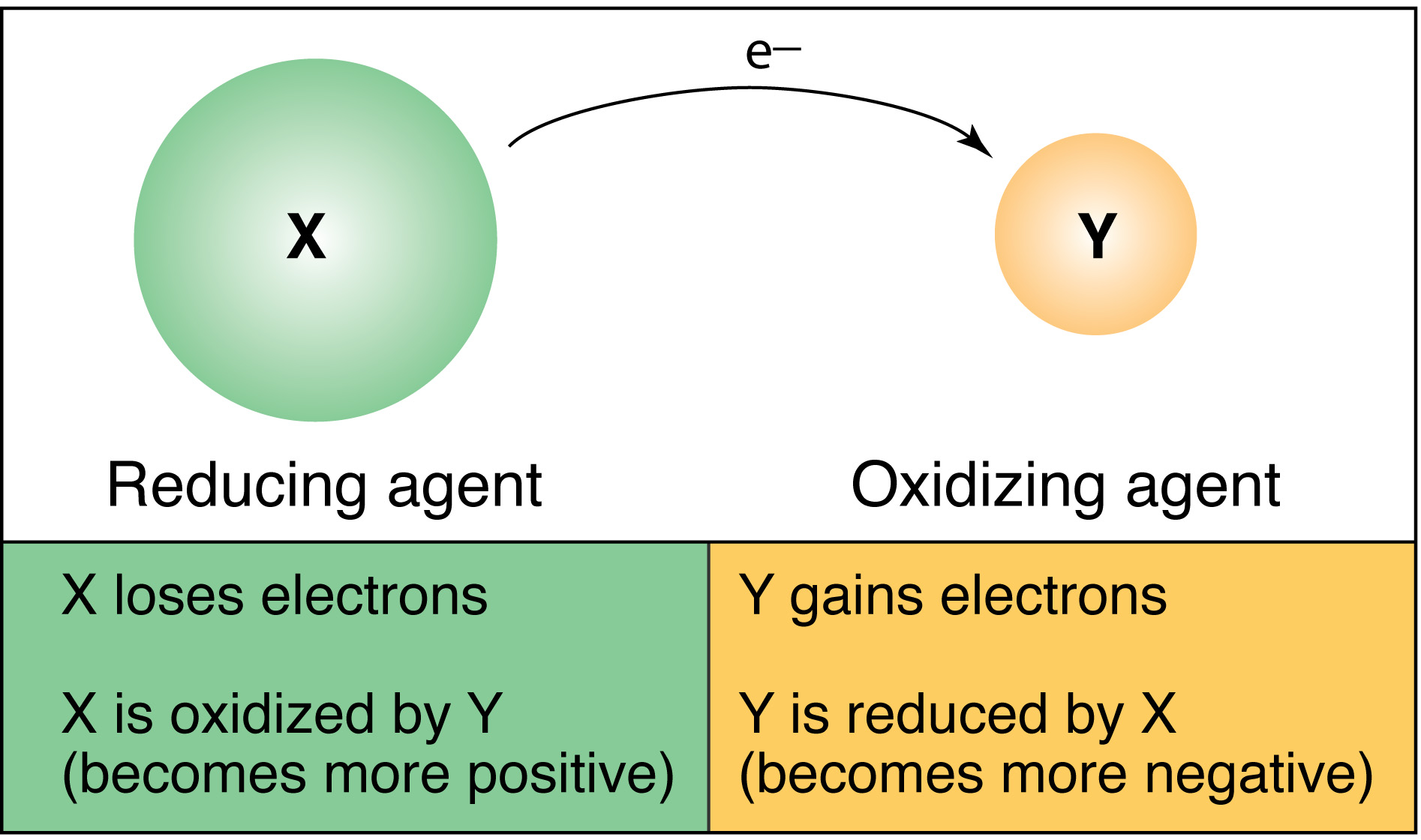
Transfer of electrons can be the driving force for a reaction. This rearrangement of atoms and electrons is what we refer to as the mechanism of the reaction. One example of a reaction in which electron transfer is the driving force is given below. Some force must move the electrons along from one point to another.
Electrons do not flow that is current doesnt exist without a driving force. Answer to What do we mean when we say that the transfer of electrons can be the driving force for a reaction. When one say that transfer of electrons can be the driving force then it means that transfer of electrons makes the reaction to happen.
8132020 That exothermic half-reaction served as part of the driving force for the electron transport chain the associated proton pumping and ATP formation. When two solutions of ionic substances are mixed and a precipitate forms what is. Calculate ΔU for that process.
Electron Transfer as a Driving Force CASE STUDY. BDFEs can be analyzed as sums of electron and proton transfer steps and can therefore be obtained from p K a and E. Achieving this state in conjunction with lowering the potential energies is a major driving force for bond formation.
Oxidation is the removal of electrons from an atom and reduction is the addition of the electrons to an atom. 8132020 In oxidationreduction redox reactions electrons are transferred from one species the reductant to another the oxidant. The driving force for a solution HAT reaction is most easily discussed as the difference in the bond dissociation free energies BDFEs of the reactants and products.
To drive that reaction backward by stripping electrons from water will require a very strong oxidising agent. This transfer of electrons provides a means for converting chemical energy to electrical energy or vice versa. For example redox reactions are what makes your batteries work.