Transfer Of Electrons Driving Force Reaction

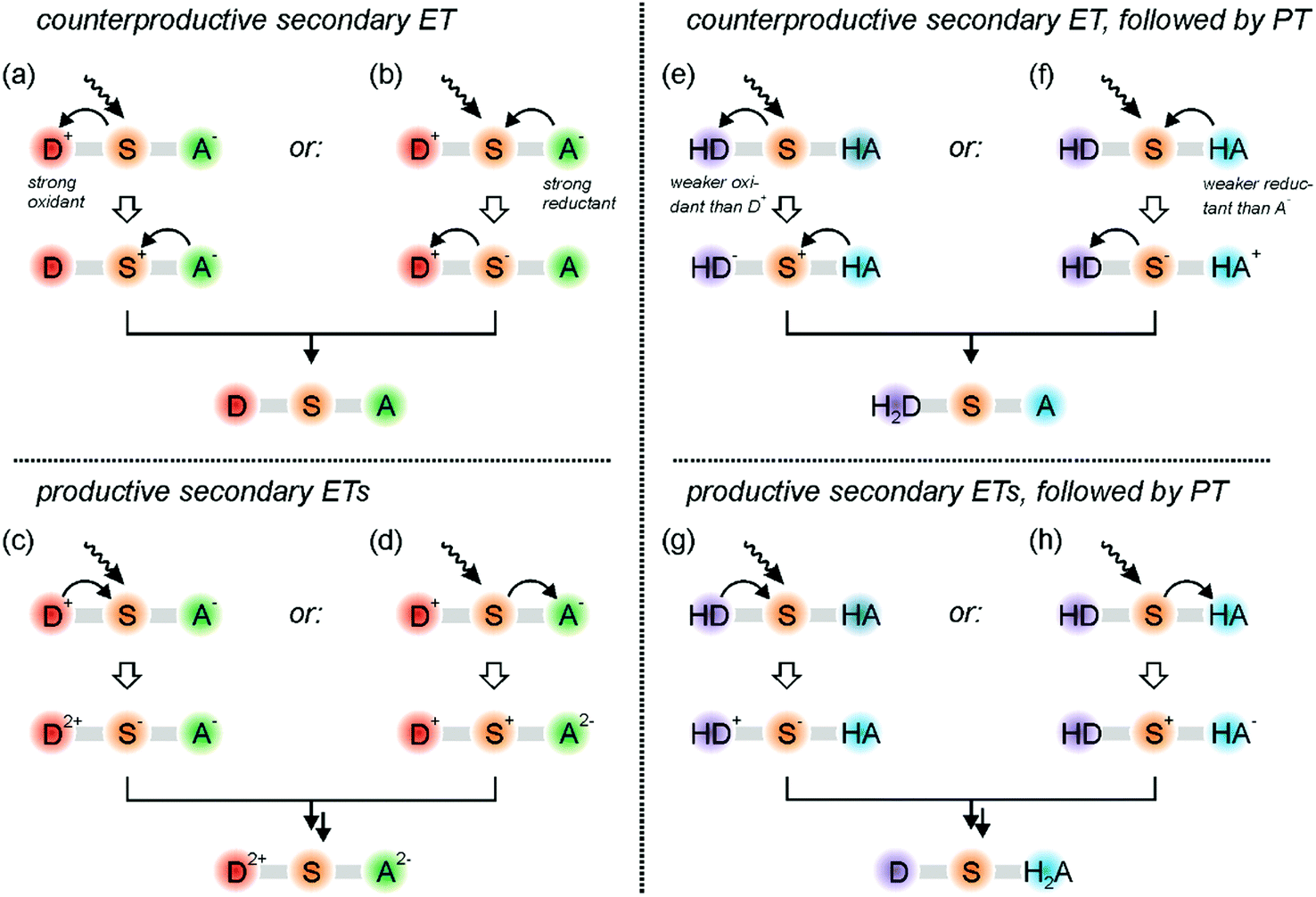
Something to remember is like attacks like.
Transfer of electrons driving force reaction. Oxidation is the removal of electrons from an atom and reduction is the addition of the electrons to an atom. Transfer of electrons formation of a gas formation of a solid or dissolving of a salt. The reaction is based on a reactivity series.
In the first reaction PQH 2 binds to the complex on the lumen side and one electron is transferred to the iron-sulfur center which then transfers it to cytochrome f which then transfers it to plastocyanin. C The transfer of electrons from N to O D The solubility of AgNO 3. General formula is ABCBAC single type of anion exchanged in the equation element displaces a charged ion in the reaction aka metal displacement not reversible driving force is transfer of electrons only occurs if A is more active than B.
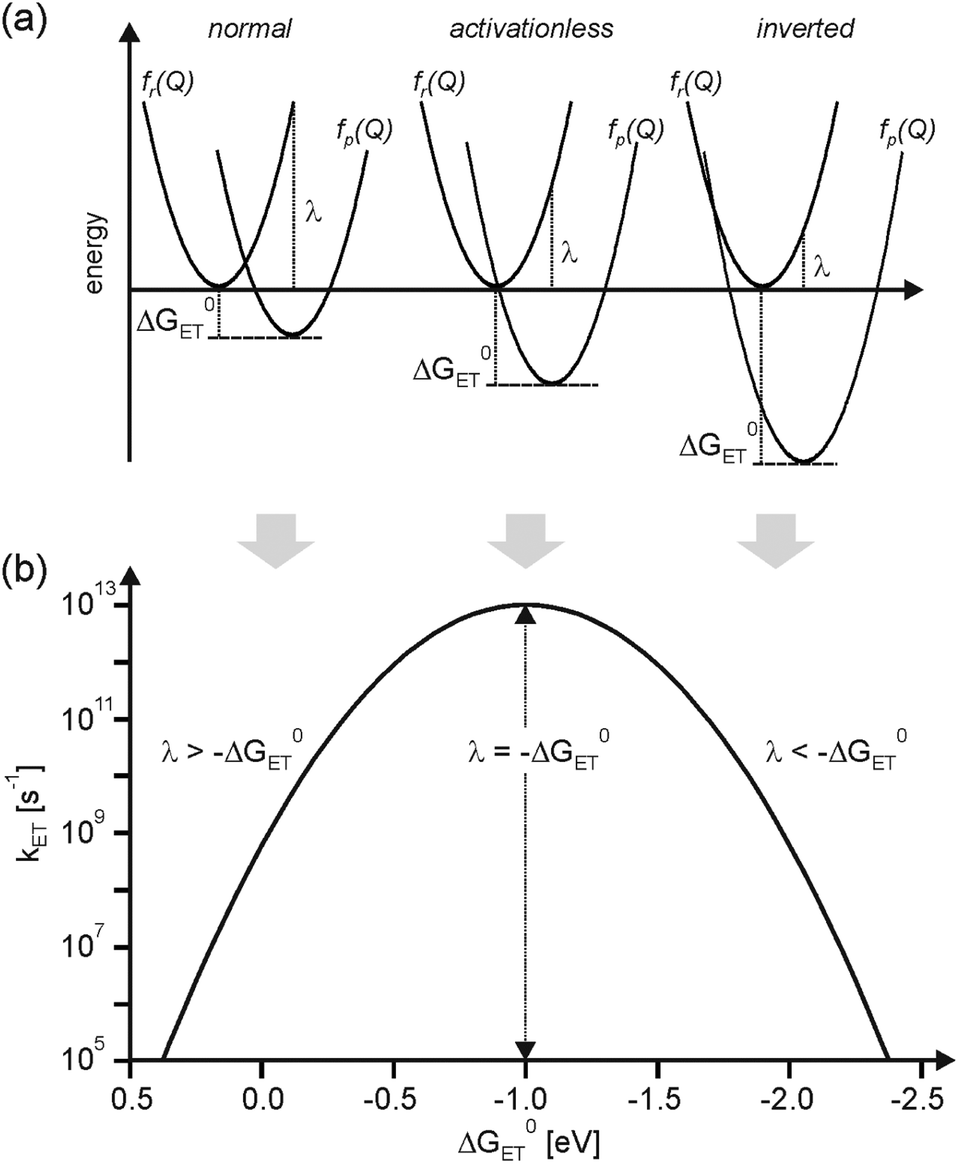
B The formation of a solid. Below is an example. The driving force for a solution HAT reaction is most easily discussed as the difference in the bond dissociation free energies BDFEs of the reactants and products.
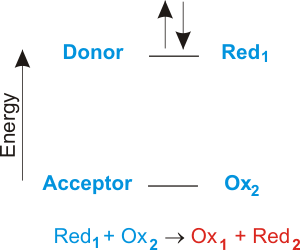
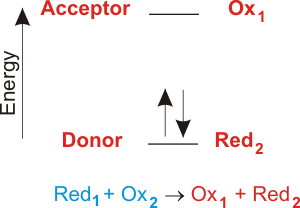
What is the difference between P4 and 4P in a chemical reaction. One example of a reaction in which electron transfer is the driving force is given below. Fundamentals of Electrochemistry Basic Concepts 2 The first two reactions are known as 12 cell reactions Include electrons in their equation 3 The net reaction is known as the total cell reaction No free electrons in its equation 4 In order for a redox reaction to occur both reduction of one compound and oxidation of another must take place simultaneously Total number of.
2 H 2 O 2 H 2 O 2 Example. For this type of reaction it it good t remember that like attacks like. The second electron is transferred to heme b L which then transfers it to heme b H which then transfers it to PQ.
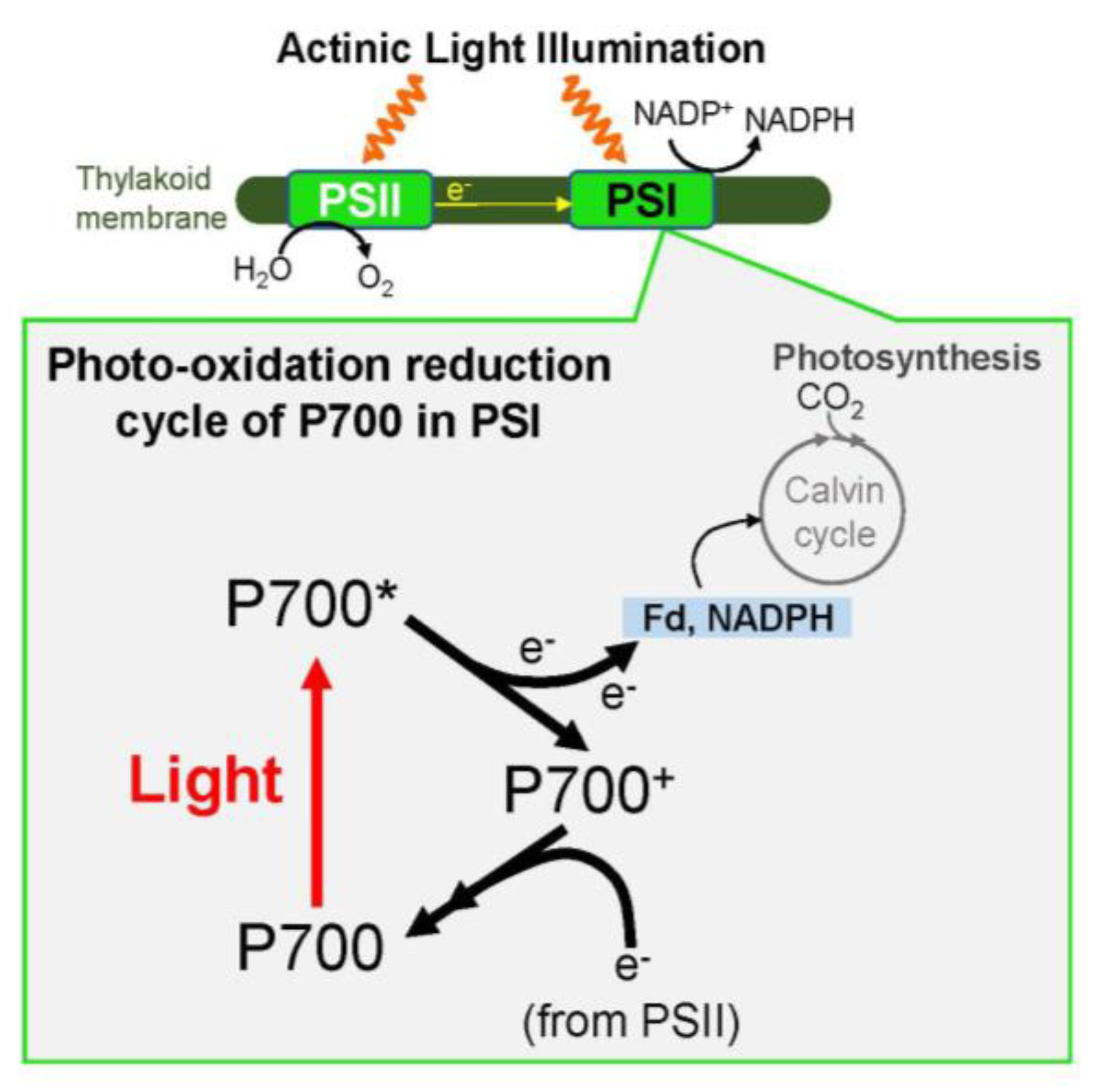
Most common driving forces are. Biological processes such as respiration photosynthesis and the breakdown of food molecules consist of sequences of electron transfer reactions that serve to transport and utilize energy from the sun. A Which of the following is not a driving force for a chemical reaction.