Transfer Of Electrons Explained

One reactant donates electrons to another reactant.
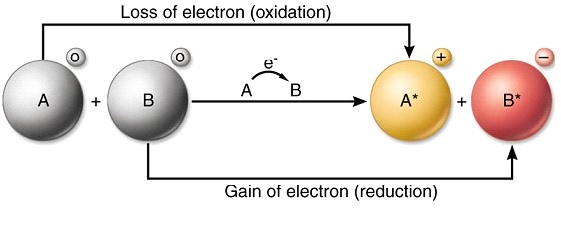
Transfer of electrons explained. These electron-transferring chemical reactions are called reduction-oxidation reactions or redox reactions for short. 8152020 In the above reaction magnesium reduces the copper II ion by transferring electrons to the ion and neutralizing its charge. 7312002 Redox reactions are characterized by the actual or formal transfer of electrons between chemical species most often with one species the reducing agent undergoing oxidation losing electrons while another species the oxidizing agent.
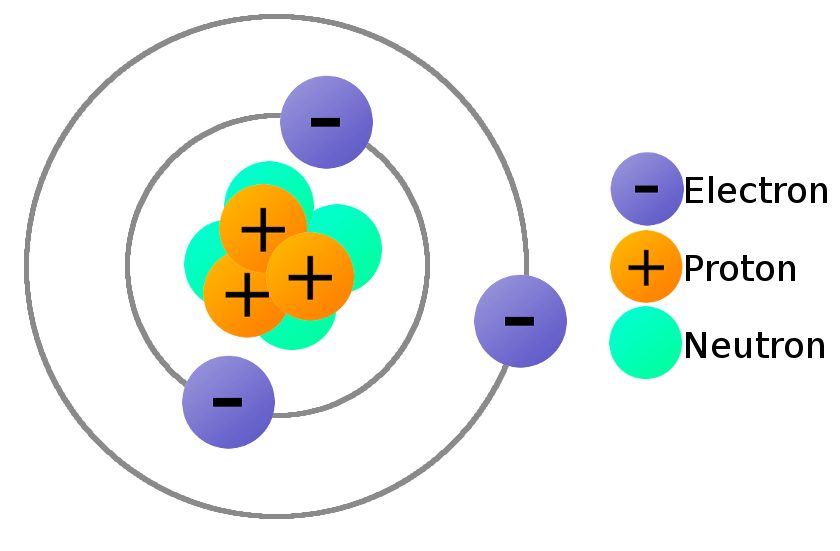
There are terms like Ionization energy Electron affinity and Electronegativity mostly used in chemistry both describe the love for the atoms towards the electrons. A Although mechanisms differ slightly they all propose the participation of a base that activates the dihydrolipoamide by abstracting a proton from the reduced state. 10172019 Whenever electrons are transferred between objects neutral matter becomes charged.
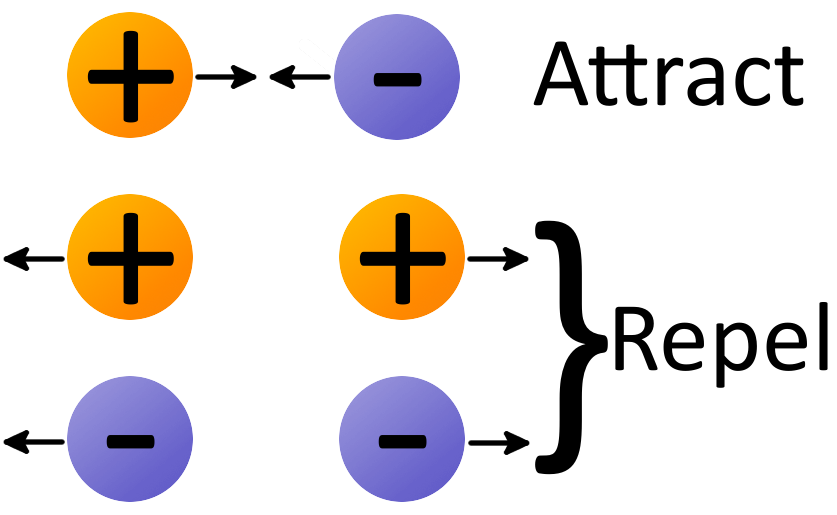
The process of efficiently and controllably moving electrons around is one of the primary regulation mechanisms in biology. The ionic bond between ions results from the electrostatic attraction of opposite charges. In each case the total charge remains the same.
Similarly cupric ions gain electrons and get reduced to copper atoms. Then the electron is transferred to the iron atom in the next cytochrome carrier in the electron transfer chain thus oxidizing the first. 11142017 An oxidation-reduction redox is a chemical reaction involving transfer of electrons between two species.
We saw this in the formation of NaCl. In the above reaction zinc atoms remove electrons and get oxidized to zinc ions. The two former ones denote the energy involved while the latter one denotes the ability for transferring electrons.
Thus there will be an electron transfer takes place between a sodium atom and a chlorine atom. The copper II ion is acting as an oxidizing agent. The transfer of electrons releases stored energy that can be used to attach inorganic phosphate to ADP to form ATP.