Transfer Of Electrons Formula
Electron Transfer The concept of oxidation arises from the combination of elemental oxygen with other elements to form oxides as in this example using aluminum.
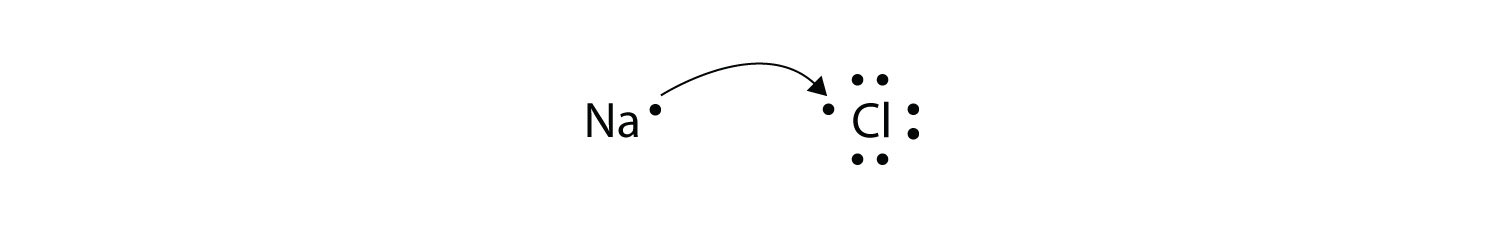
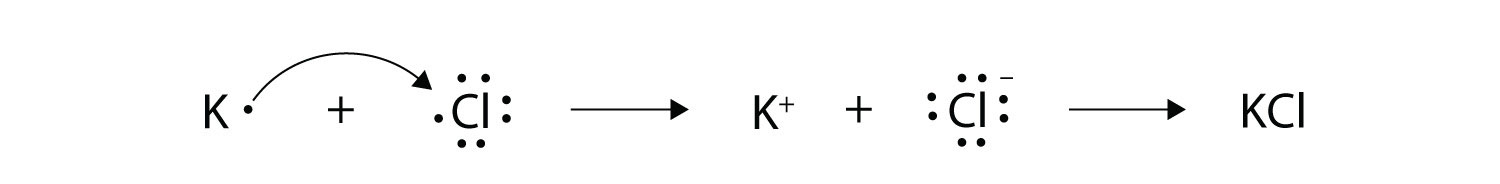
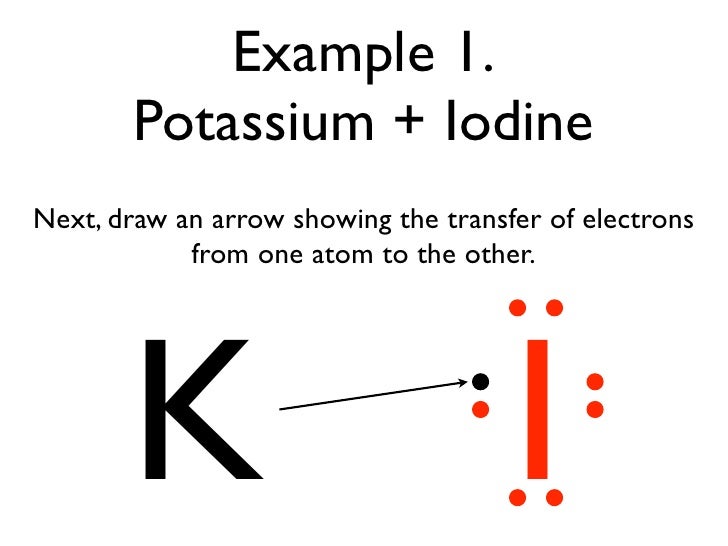
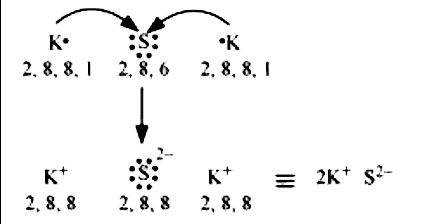
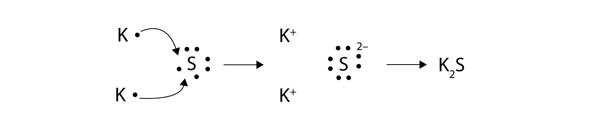
Transfer of electrons formula. The two ions each have octets as their valence shell and the two oppositely charged particles attract making an ionic bond. Thus the charge of a cation formed by the loss of all valence electrons is. The two ions each have octets as their valence shell and the two oppositely charged particles attract making an ionic bond.
We saw this in the formation of NaCl. Differential Pulse Polarography Electron Transfer. 4212012 Since the reaction shows 6 bromine atoms involved and armed with the knowledge that bromine atoms gain 1 electron each we can surmise that 6 electons 3 from each of the 2 scandium atoms are transferred.
The periodic table can help us recognize many of the compounds that. 4 sodium nitride Na and N Formula 6 magnesium phosphide Mg and P Formula S 36 2 carbon tetrabromide CBra 3 dihydrogen monoxide H20 Final Answer. 4Al 3O 2 2Al 2 O 3.
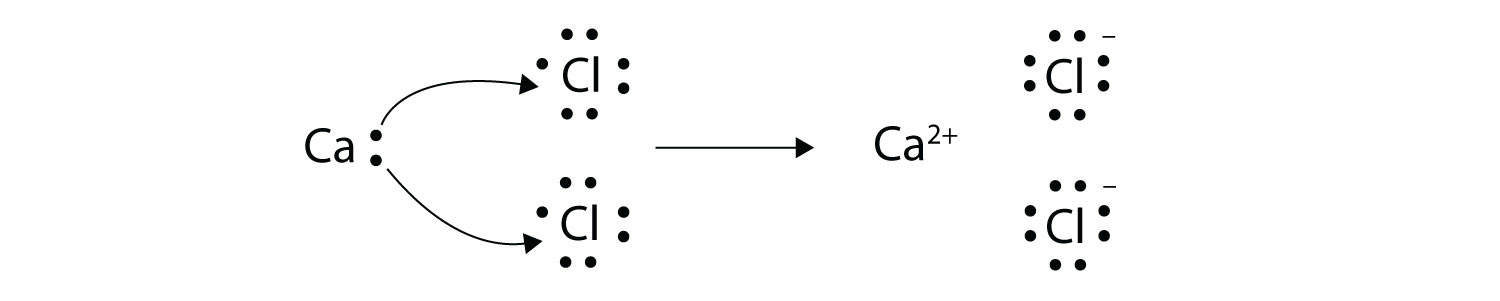
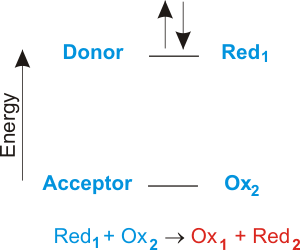
The other reason to study electron transfer is that it is a very simple kind of chemical reaction and in understanding it we can gain insight into other kinds of chemistry and biochemistry. 10192018 Similarly each calcium atom group 2 can give up two electrons and transfer one to each of two chlorine atoms to form CaCl 2 which is composed of Ca 2 and Cl ions in the ratio of one Ca 2 ion to two Cl ions. 2 calcium chloride Ca and Cl 4 sodium nitride Na and N Formula.
A compound that contains ions and is held together by ionic bonds is called an ionic compound. In mathematical terms Restricted linear energy transfer is defined by L Δ d E Δ d x displaystyle L_Delta frac textdE_Delta textdx where d E Δ displaystyle textdE_Delta is the energy loss of the charged particle due to electronic collisions while traversing a distance d x displaystyle textdx excluding all secondary electrons with kinetic energies larger than Δ. The Law of conservation of charge states that the net charge of an isolated system remains constant.
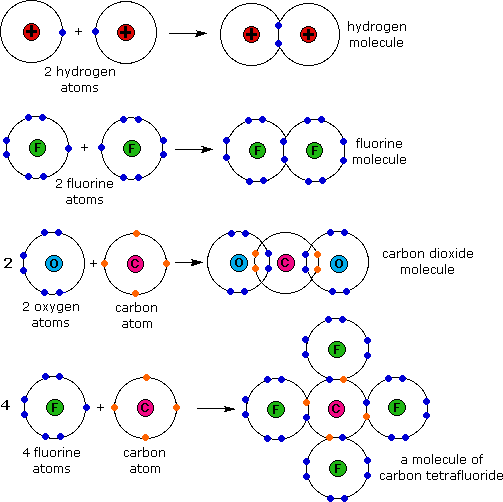
For the formation of Na2O. An electron dot formula shows how electrons are shared between two or more atoms with the use of dots. We saw this in the formation of NaCl.