Transfer Of Electrons From Metal To Nonmetal Results In The Formation Of
2252002 In the simplest case the cation is a metal atom and the anion is a nonmetal atom but these ions can be of a more complex nature eg.
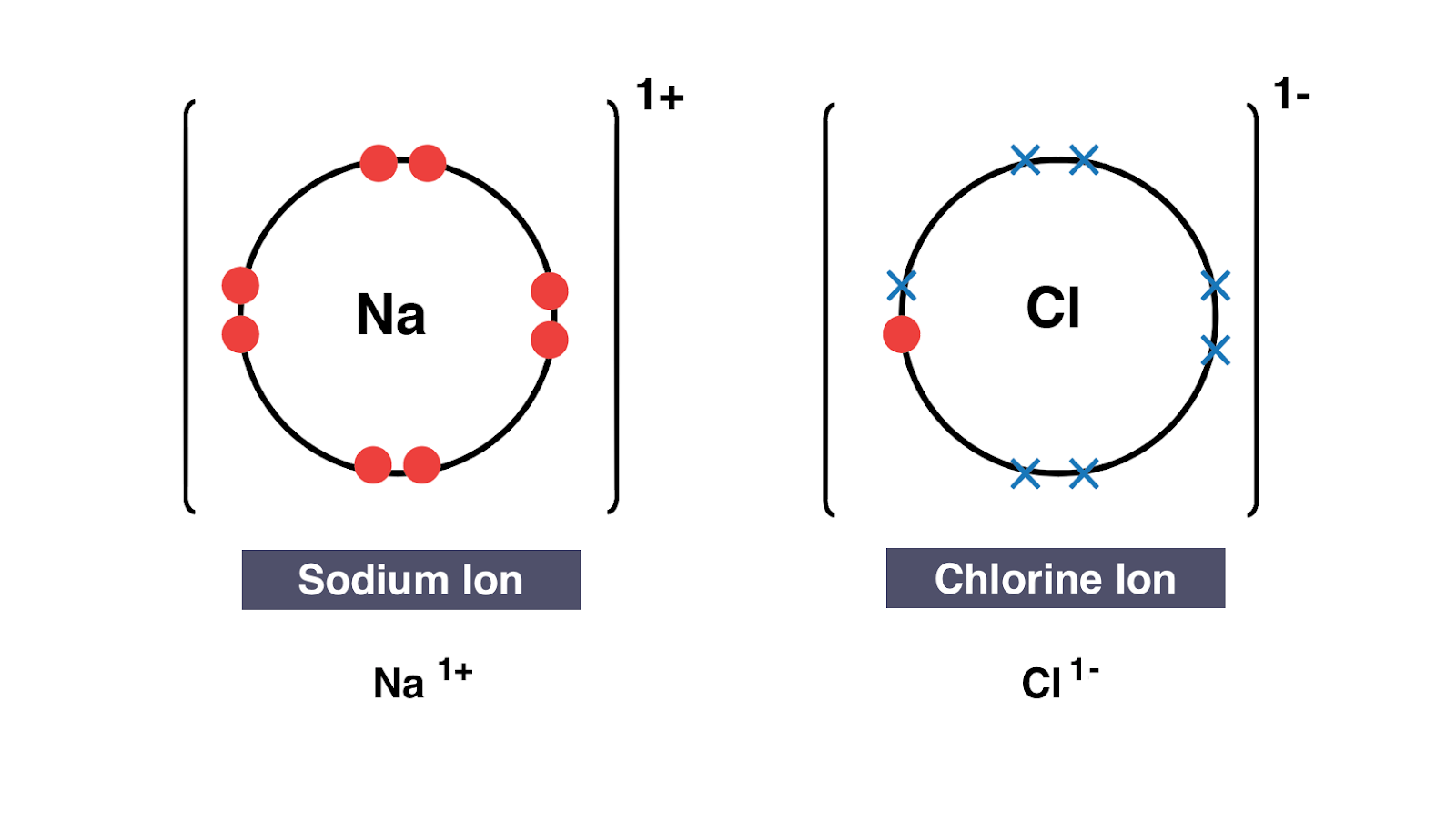
Transfer of electrons from metal to nonmetal results in the formation of. A cation a positive ion forms when a neutral atom loses one or more electrons from its valence shell and an anion a negative ion forms when a neutral atom gains one or more electrons. In reality electron density remains shared between the constituent atoms meaning all bonds have some covalent character. This creates oppositely charged ions.
Ionic Bonding and Electron Transfer Ions are atoms or molecules bearing an electrical charge. We saw this in the formation of NaCl. I They are the solids having high melting point.
The noble gas involved is the one following the nonmetal in the p 3. I State the Type of Bonds in Y. Iii Will It Be a Good Conductor of Electricity.
Ii They are soluble in water. In covalent bonding the two electronsshared by the atoms are attracted to the nucleus of both atoms. When a metal reacts with a non-metal electrons transfer from the metal to the non-metal.
Select all that apply. 9162014 Ionic bonds are caused by electrons transferring from one atom to another. A similar process occurs between Mg atoms and O atoms except in this case two electrons are transferred.
Metal atoms lose electrons to. 9132020 Ionic bonding is the complete transfer of valence electron s between atoms. Show formation of K 2 O and MgO by transfer of electrons Share with your friends.



















