Transfer Of Electrons From One Material To Another

There are three different ways to transfer electrons from one object to another.
Transfer of electrons from one material to another. Quantization of charge means that when we say something has a given charge we mean that that is how many times the charge of a single electron it has. The laws of mechanics and electricity would. ˈ r ɛ d ɒ k s redoks or ˈ r iː d ɒ k s reedoks is a type of chemical reaction in which the oxidation states of atoms are changed.
The material by electrification the transfer of electrons from one body to another takes placeand the atom is. Redox reactions are characterized by the actual or formal transfer of electrons between chemical species most often with one species the reducing agent undergoing oxidation losing electrons. As electrons jump through the air they produce intense light and heat.
Lewis formulation of an ionic bond stems from the transfer of electrons from one atom to another. 492020 If we put two different materials in contact and one attracts electrons more than the other its possible for electrons to be pulled from one of the materials to the other. Electric charge is the transfer of electrons from one material to another.
In charging by rubbing electrons move from one material to another because. 7312002 Redox reductionoxidation pronunciation. Ionic bonding is the complete transfer of valence electron s between atoms.
During this friction the surface atoms of both the materials are pressed together bringing the electron clouds to a closer proximity thereby causing an interaction which results in the transfer of electrons among the clouds. Soon electrons jump from the cloud to the Earth. So negative charge means that there is an excess of electrons while positive charge is a deficiency of electrons.
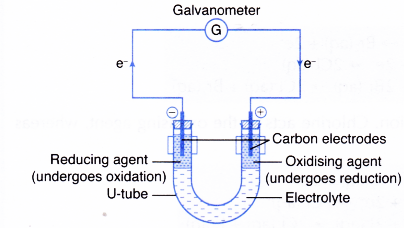
It is a type of chemical bond that generates two oppositely charged ions. 1212020 Redox Reactions by Transfer of Electrons at a Distance In all redox reactions electrons are transferred from the reducing agent to the oxidising agent. Transfer of electrons from one uncharged to another by rubbing the objects together induction the transfer of electrons from one object to another without the two objects touching.