Transfer Of Electrons From One Species To Another

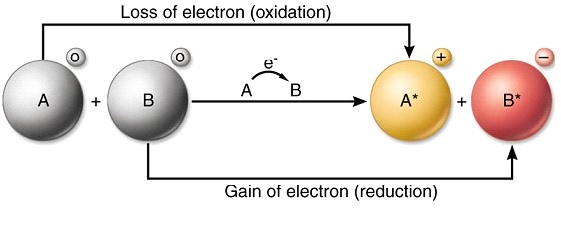
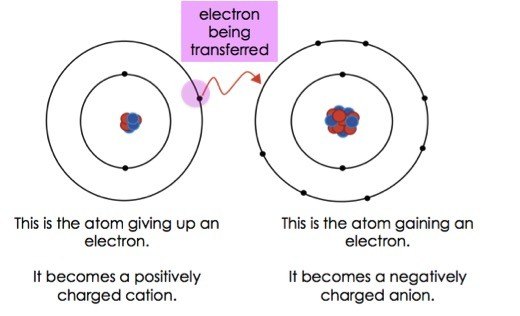
In electron transfer the number of electrons lost must equal the number of electrons gained.
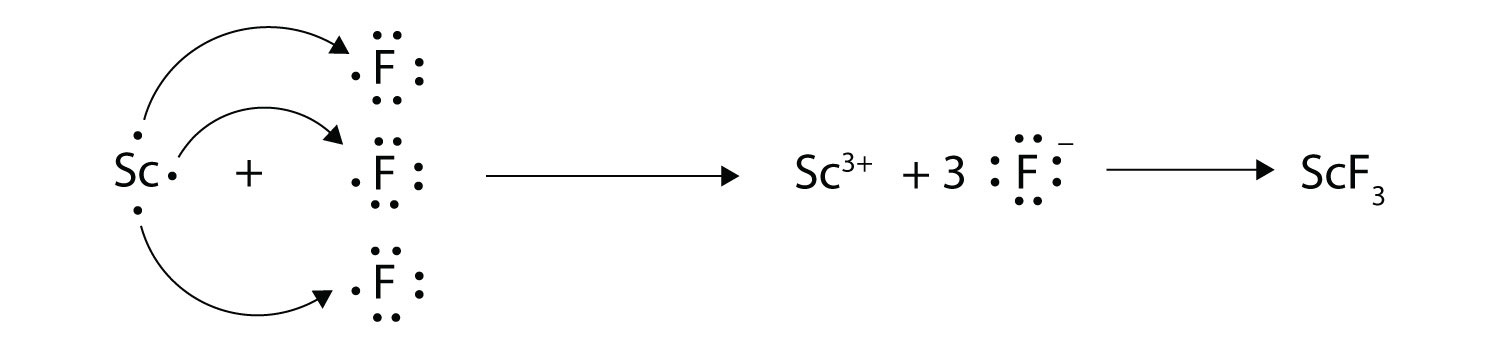
Transfer of electrons from one species to another. There are two major types of electrochemical cells. In electron transfer the number of electrons lost must equal the number of electrons gained. Electron transfer potentials depend on the chemical species involved in the reaction.
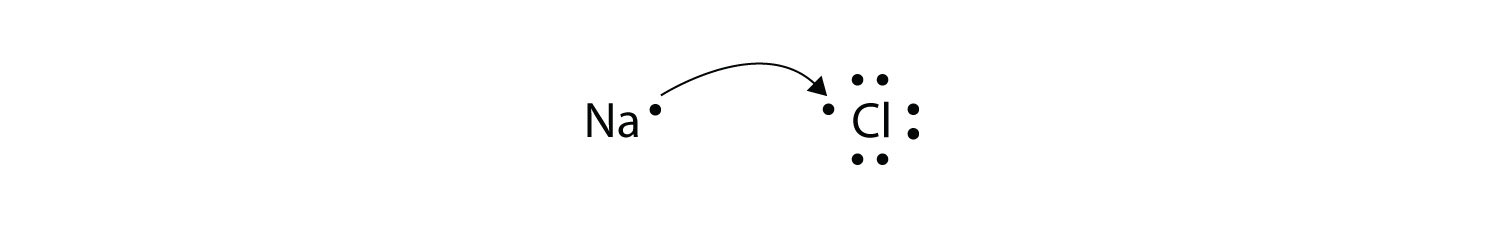
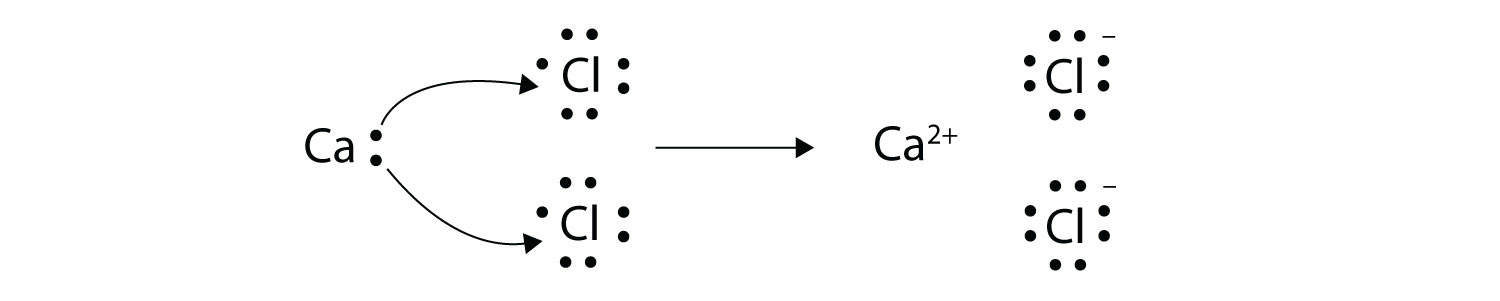
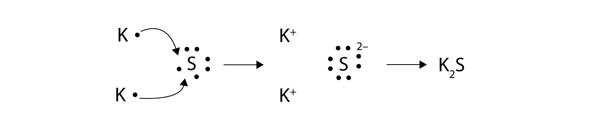
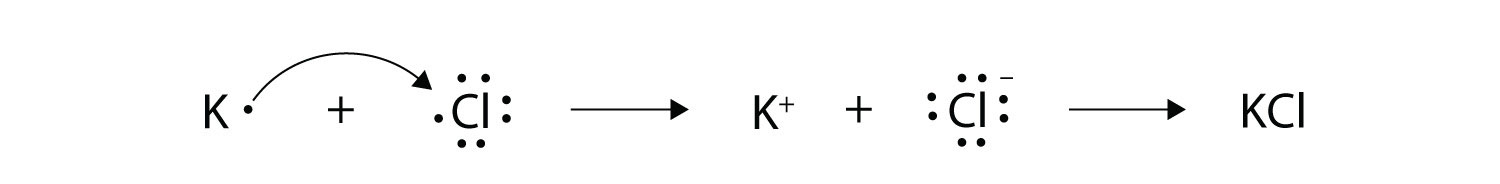
Voltaic also galled galvanic and electrolytic. Ionic bonds are caused by electrons transferring from one atom to another. We saw this in the formation of NaCl.
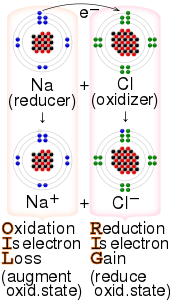
The process of efficiently and controllably moving electrons around is one of the primary regulation mechanisms in biology. And the species that gain electrons is said to be reduced. ___ bonds result from the transfer of electrons from one element to another ionic bonds ___ are discrete groups of atoms that are held together by covalent bonds.
The sparks flying outwards are globules of molten iron trailing smoke in their wake. Oxidation-reduction reactions also called redox reactions involve the transfer of electrons from one species to another. We saw this in the formation of NaCl.
The chemical species being oxidized loses electrons to. 6182019 Oxidation-reduction reactions also called redox reactions involve the transfer of electrons from one species to another. When the reducing and oxidising agents are mixed together as in the previous reactions the transfer of electrons occurs quickly and cannot be detected.
The reaction that occurs when iron steel wool is placed in a solution of CuSO 4 is shown in the table below. In these chemical systems the species that loses electrons is said to be oxidized and the species that gain electrons is said to be reduced. 972011 Oxidationreduction reactions involve the transfer of electrons from one species to another oxidising one species and reducing the other.