Transfer Of Electrons In A Battery

The rate at which this electron transfer occurs affects the efficiency of the battery.
Transfer of electrons in a battery. Each of these is called a half reaction. Ions are electrically charged atoms that have lost or gained electrons. 1202021 The battery stores or releases energy as the fluids flow through the working part of the battery.
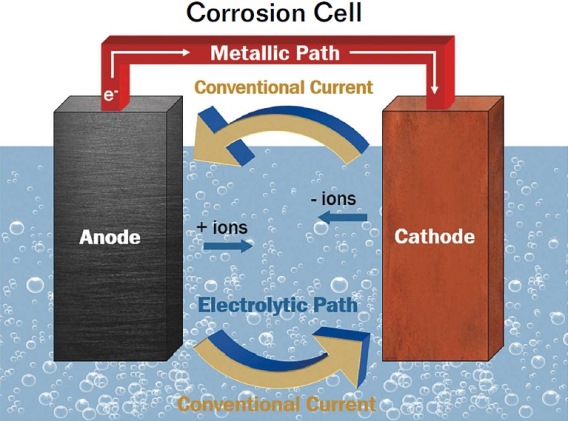
The current in the battery arises from the transfer of electrons from one electrode to the other. Describe the transfer. Mass transfer of electrons from zinc plate The number of electrons on the zinc plate increases the more the zinc breaks down.
During discharging the oxidation reaction at the anode generates electrons and reduction reaction at the cathode uses these electrons and therefore during discharging electrons flow from the anode to the cathode. But the electrolyte keeps the electrons from going straight from the anode to the cathode within the battery. 1212021 The battery stores or releases energy as the fluids flow through the working part of the battery.
5232007 Depends on the size of the battery and how much material is inside the battery. The electrode from which electrons are removed becomes positively charged while the electrode to which they are supplied has an excess of electrons and a negative charge. This lithium atom is highly unstable and will instantly form a lithium ion and an electron.
The rate at which this electron transfer occurs affects the efficiency of the battery. When the electrons move through the filament they experience high resistance. Redox reactions can be used in electrochemical cells to produce electricity.
3192005 Oxidationreduction reactions are electron-transfer reactions. In a conventional battery such as lithum-ion atoms in its electrodes switch from positive to negative charge and vice versa as they lose or gain electrons when the. The electrons just circle around the circuit.