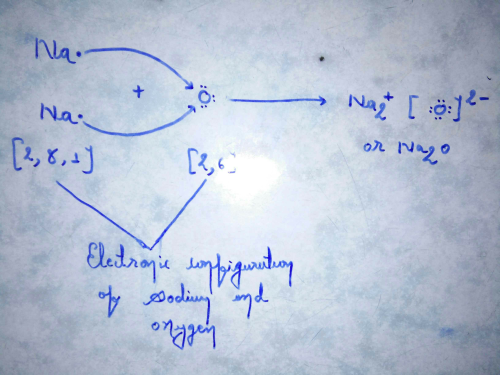
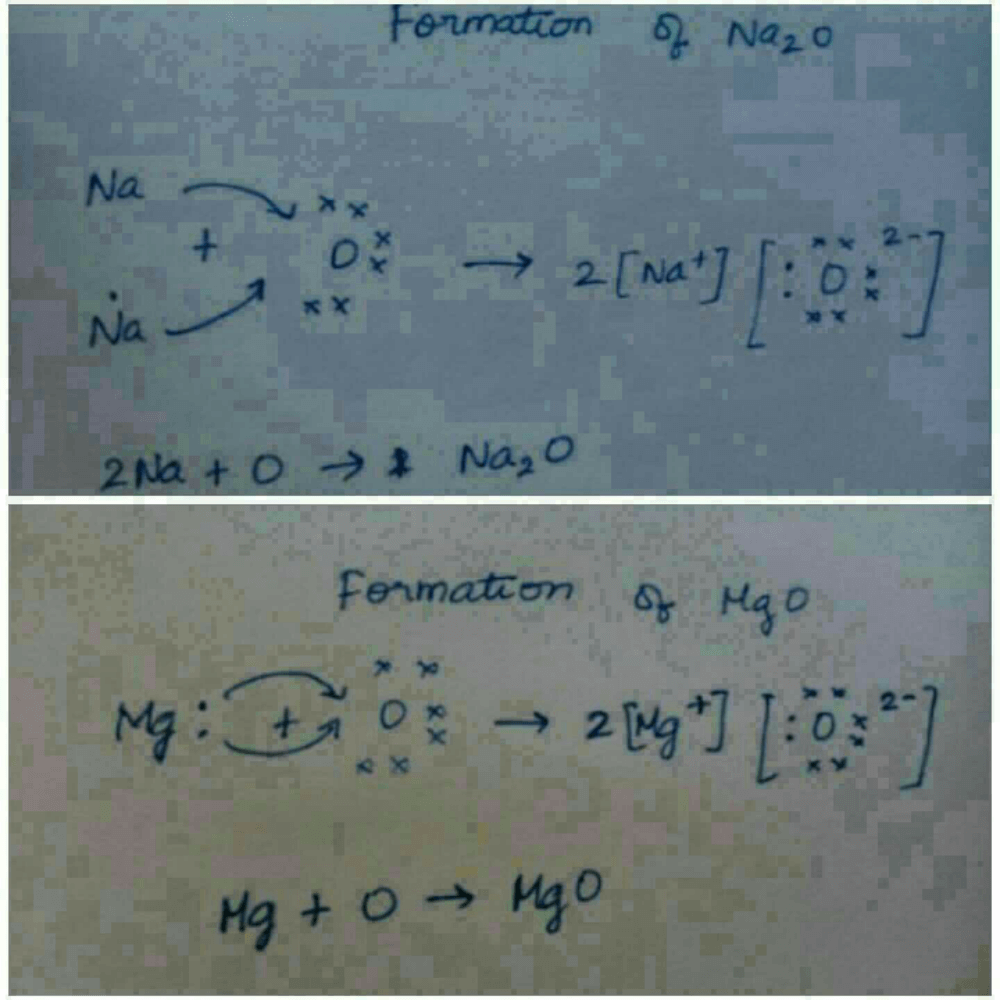
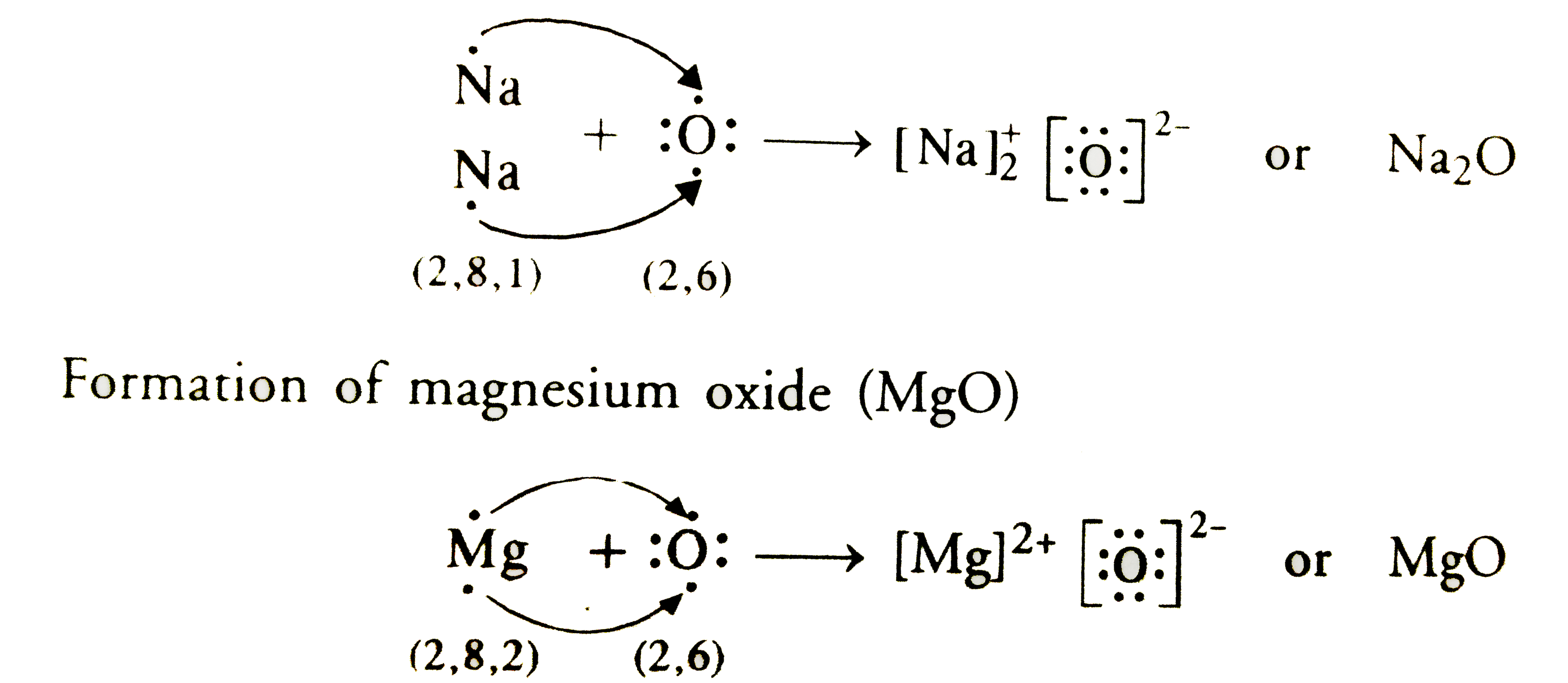
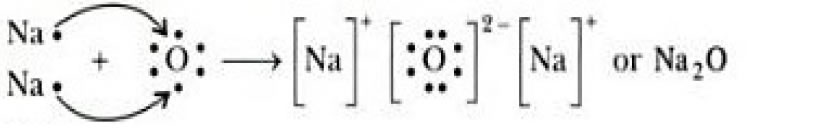Transfer Of Electrons In Na2o

So the magnesium atom donates its 2 valence electrons and forms a stable magnesium ion Mg2 to attain the electronic arrangement of neon gas.
Transfer of electrons in na2o. We saw this in the formation of NaCl. Na2O is therefore an ionic or electrovalent compound. Knockout JEE Main April 2021 One Month Personalized AI Tutor and Adaptive Time Table Self Study Material Weekend Live Classes Mentorship from our Experts Unlimited Mock Tests and Personalized Analysis Reports 24x7 Doubt Chat Support.
A Write down the electron arrangement in i a magnesium atom and ii a chlorine atomb How many electrons are there in the valence shell of i a magnesium atom and ii a chlorine atomc Show the formation of magnesium chloride from magnesium and chlorine by the transfer of electronsd State whether magnesium chloride will conduct. Sodium has one valance electron whereas oxygen has 6. In electron transfer the number of electrons lost must equal the number of electrons gained.
C Show on a diagram the transfer of electrons between the atoms in the formation of MgO. A similar process occurs between Mg atoms and O atoms except in this case two electrons are transferred. Show the formation of Na2 Oby the transfer of electrons.
You may need to download version 20 now from the Chrome Web Store. Write the electron arrangement in i a magnesium atom and ii an oxygen atom. Ii Show the formation of Na 2 O and MgO by the transfer of electrons.
How many electrons are there ini the valence shell of i a magnesium atom and ii an oxygen atom. Sodium will lose the one valance electron to full fill his octet whereas oxygen will gain this one electron but to complete the octet of oxygen it needs two electrons. Iii What are the ions present in these compounds.
Learn about the structure properties and uses of sodium oxide here. Sodium Oxide Na2O - Sodium oxide is an inorganic compound with the chemical formula Na2O. Ii Show the formation of Na2O and MgO by the transfer of electrons.