Transfer Of Electrons Meaning

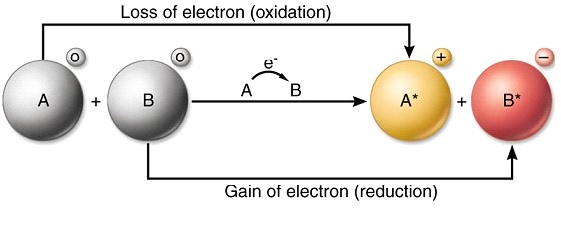
In the aluminumoxygen example the aluminum was oxidized and the oxygen was reduced because every electron transfer reaction involves simultaneous oxidation and reduction.
Transfer of electrons meaning. During this friction the surface atoms of both the materials are pressed together bringing the electron clouds to a closer proximity thereby causing an interaction which results in the transfer of electrons among the clouds. In an electron transfer reaction an element undergoing oxidation loses electrons whereas an element gaining electrons undergoes reduction. Due to the transfer of electrons.
2Covalent bond- Some electrons from each atom are shared by all the atoms such that those electrons complete the octet of each contributing atom. Oxidation is a process in which an atom or a group of atoms taking part in chemical reaction loses one or more electrons. We saw this in the formation of NaCl.
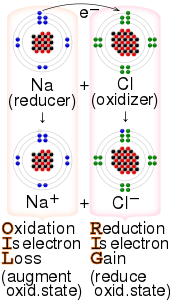
The process of transference of electrons is described as redox process. We saw this in the formation of NaCl. The charged species are held together by electrostatic force of attraction.
In electron transfer the number of electrons lost must equal the number of electrons gained. Nutritional requirements is generated by means of electron-transfer reactions in which electrons move from an organic or inorganic donor molecule to an acceptor molecule via a pathway that conserves the energy released during the transfer of electrons by trapping it in a form that the cell can use for its chemical or. The process of efficiently and controllably moving electrons around is one of the primary regulation mechanisms in biology.
Redox reaction is a type of chemical reaction. Thus there will be an electron transfer takes place between a sodium atom and a chlorine atom. Electron transfer or sharing.
10182020 Ionic bonds are caused by electrons transferring from one atom to another. In electron transfer the number of electrons lost must equal the number of electrons gained. 1 Ionic bond- some chemical species donate electrons and some accept electrons.



.PNG)