Transfer Of Electrons Occur

2252012 Flow of electrons in producing and using up energy Edexcel Biology Glossary B1 Help.
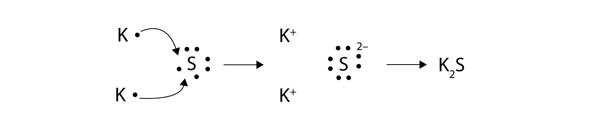
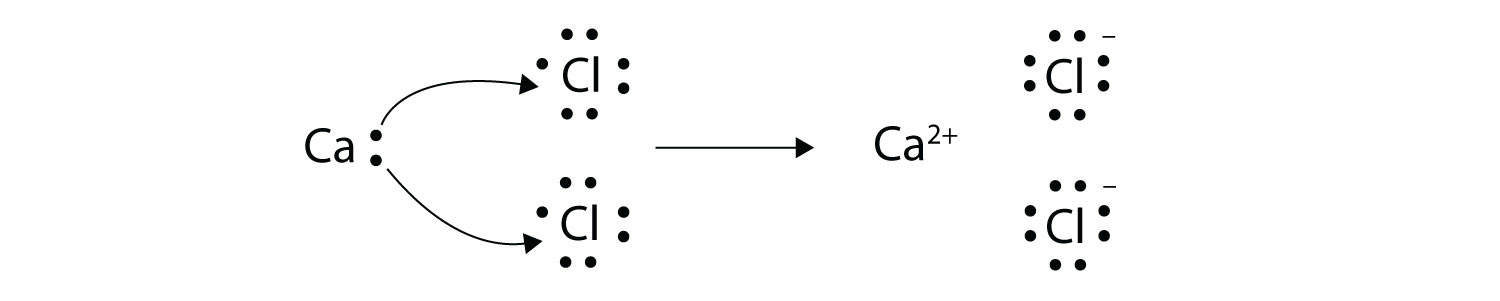
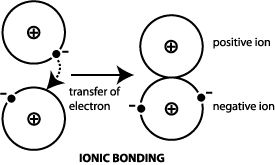
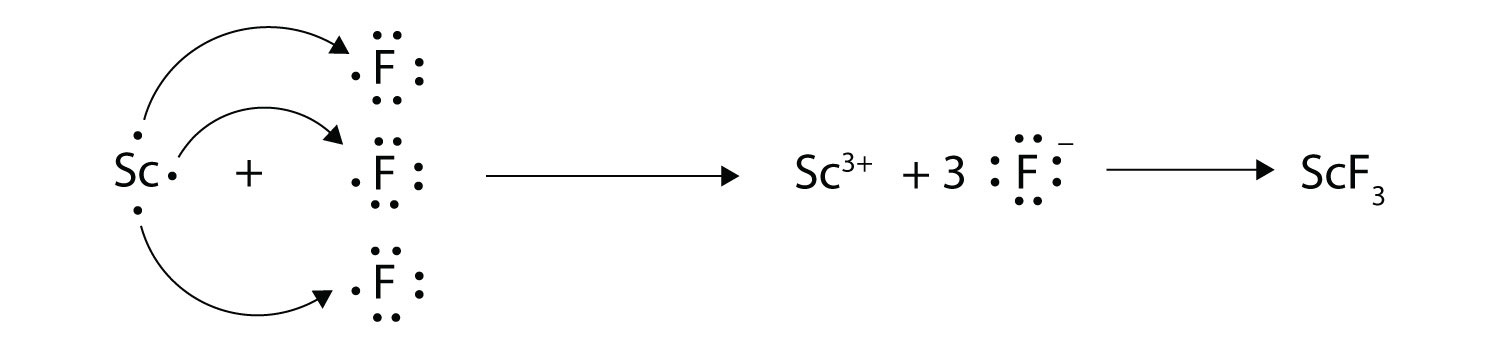
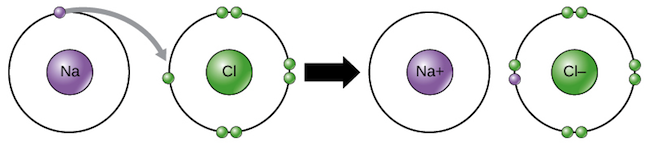
Transfer of electrons occur. 10292018 The triboelectric effect refers to the transfer of charge electrons from one object to another when they are rubbed together via friction. Link between emf of a cell and disproportionation. 1272021 Ionic bonds are caused by electrons transferring from one atom to another.
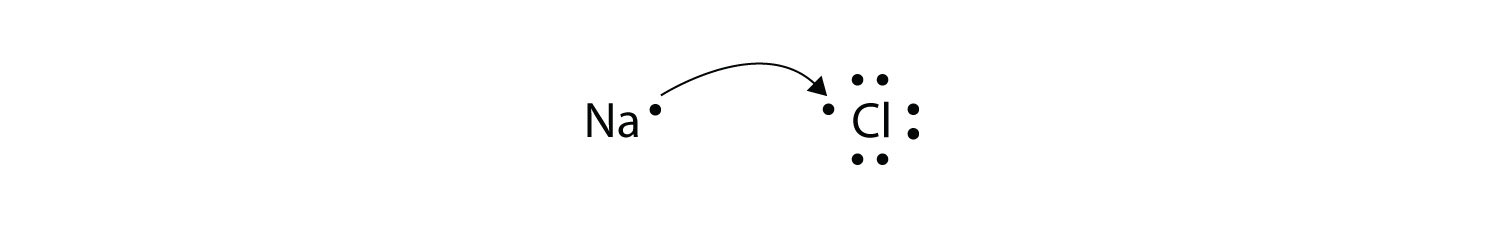
We saw this in the formation of NaCl. Krebs Cycle Question -- Need help Chemistry help. Redox reactions are characterized by the actual or formal transfer of electrons between chemical species most often with one species the reducing agent undergoing oxidation losing electrons.
As opposed to ionic bonding in which a complete transfer of electrons occurs covalent bonding occurs when two or more elements share electrons. The process of efficiently and controllably moving electrons around is one of the primary regulation mechanisms in biology. Product formation occurs through the transfer of electrons.
Because both of the nonmetals will want to _____ electrons the elements involved will share electrons. This most commonly occurs when two nonmetals bond together. 10172019 Whenever electrons are transferred between objects neutral matter becomes charged.
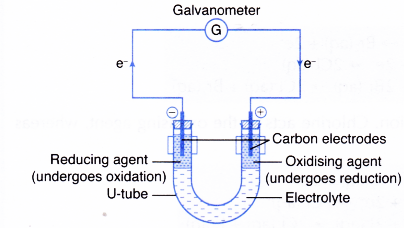
Unequal number of electrons and protons. If the solution of the oxidising agent and the solution of the reducing agent are separated by an electrolyte in a U-tube the redox reaction will still take place but the transfer of electrons will occur through the connecting wire. During electron transfer an electron is accepted by an iron atom in the pigment portion of a cytochrome molecule which thus is reduced.
1212020 The transfer of electrons occurs from the reducing agent to the oxidising agent through the connecting wires. Covalent bonding occurs because the atoms in the compound have a similar tendency for electrons generally to gain electrons. We saw this in the formation of NaCl.