Transfer Of Electrons Terms

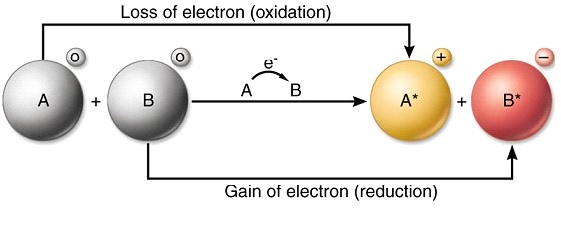
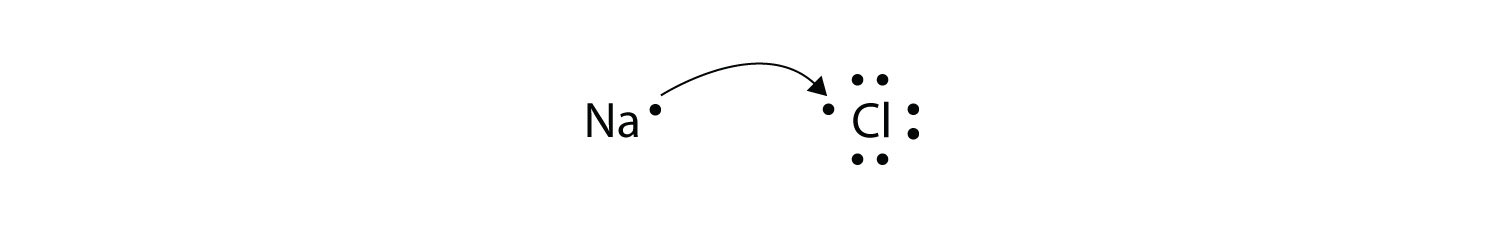
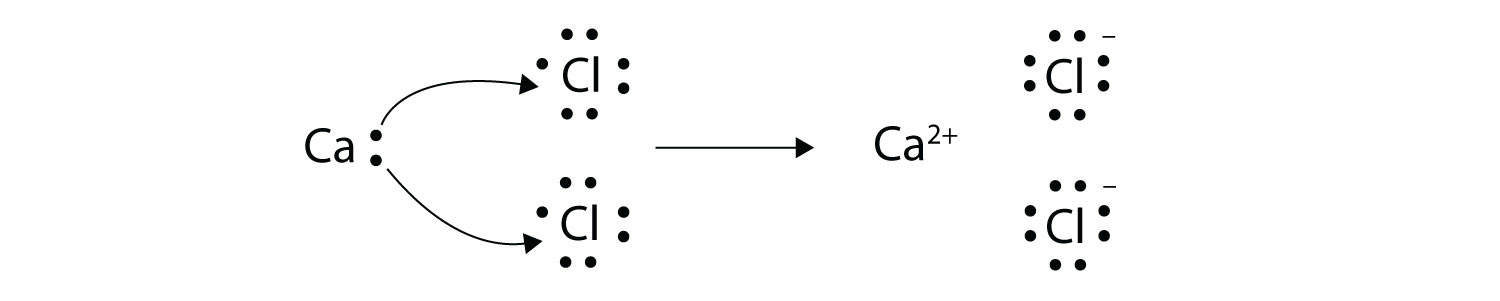
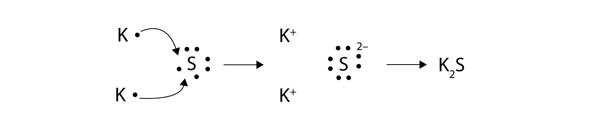
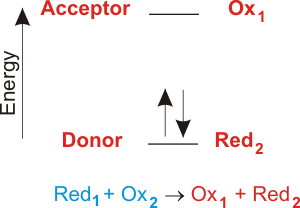
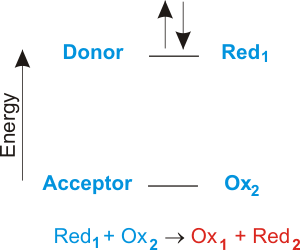
In an electron transfer reaction an element undergoing oxidation loses electrons whereas an element gaining electrons undergoes reduction.
Transfer of electrons terms. NAD is the oxidized form of the niacin and NADH is the reduced form after it has accepted two electrons and a proton. Oxidation and reduction in terms of electron transfer The commonest reaction to discuss here is the one between magnesium and copper II oxide. In the aluminumoxygen example the aluminum was oxidized and the oxygen was reduced because every electron transfer reaction involves simultaneous oxidation and reduction.
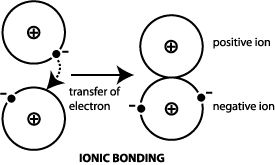
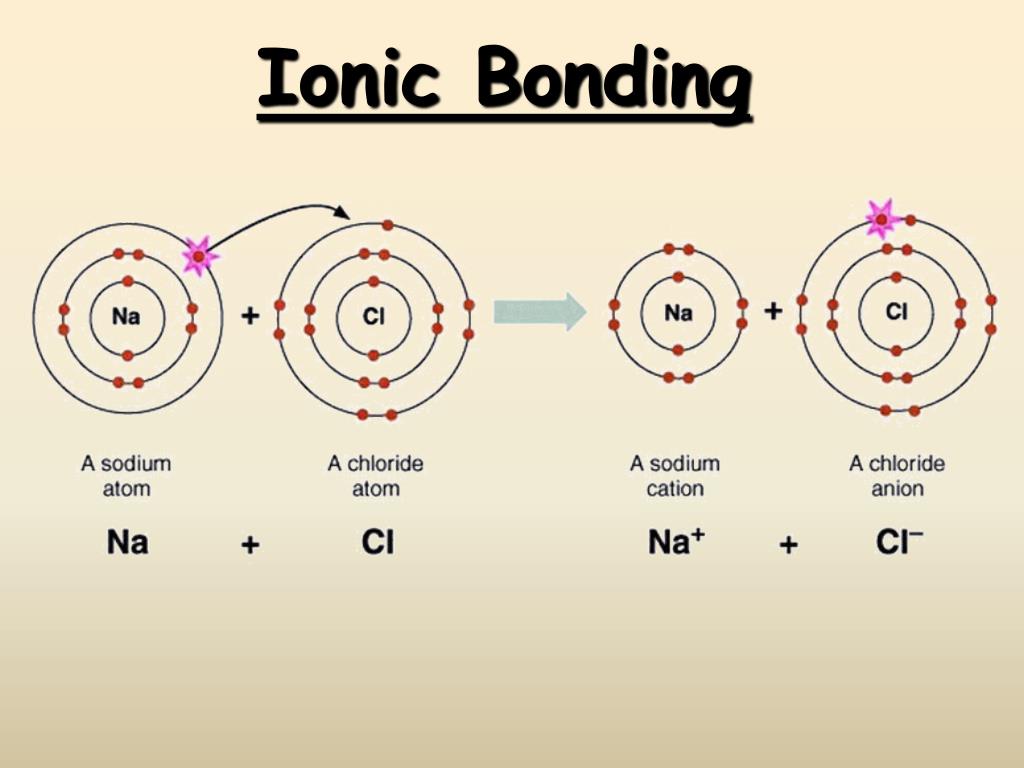
During electron transfer an electron is accepted by an iron atom in the pigment portion of a cytochrome molecule which thus is reduced. For example when atoms lose or gain electrons they become charged particles called ions. For instance consider the reaction of zinc and copper ions given below.
You will remember that this is a violent reaction in which the magnesium takes the oxygen away from the copper. In this process a body with one type of charge produces the same type of charge on a conductor. The electron transport chain ETC is a series of complexes that transfer electrons from electron donors to electron acceptors via redox both reduction and oxidation occurring simultaneously reactions and couples this electron transfer with the transfer of protons H ions across a membraneThe electron transport chain is built up of peptides enzymes and other molecules.
Electron transfer between ions and molecules at thermal energies is subject to energy resonance and FranckCondon effects. When small ions are involved electron transfer is generally inefficient unless there exists a near-resonance between an energy level of the product ion and an available recombination energy of the reactant ion. Thus reduction takes place and oxygen gets reduced.
Let us define oxidation and reduction in terms of electrons. 10172019 Whenever electrons are transferred between objects neutral matter becomes charged. The protein complex involved in these transfers is called complex III or the cytochrome b-c1 complex.
Reduction is gain of electrons. Example - 2Mgs O2 g 2MgOs In the above example each atom of oxygen gains two electrons and forms two O-2 anions. Mg CuO MgO Cu.