Transfer Of Electrons To Molecules Is Called

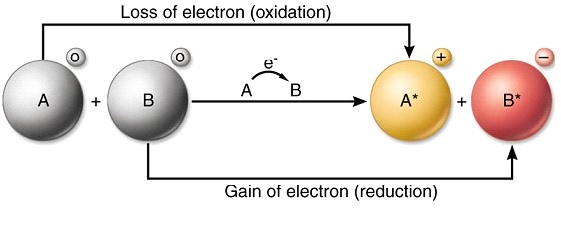
The atom that loses electrons becomes a positively charged ion.
Transfer of electrons to molecules is called. The electrons then flow through the connecting wires to the oxidising agent. Links between atoms due to the sharing or transfer of electrons are called - bonds. The NADH and FADH2 molecules that are used during the reactions of the electron transport chain are derived from glycolysis the preparatory reactions and the citric acid cycle.
In electron transfer the number of electrons lost must equal the number of electrons gained. Next the two electrons are transfered to ubiquinone. Coupled with this transfer is the pumping of one hydrogen ion for each electron.
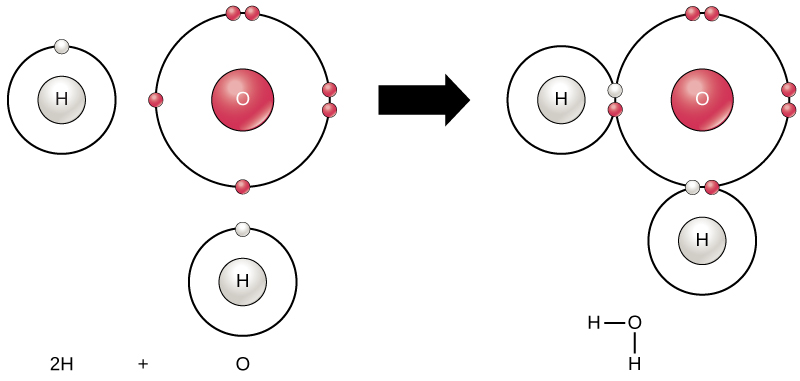
The transfer of electrons is the movement of electrons from one atom to another atom. 11112020 The electron transport chain ETC is a group of proteins and organic molecules found in the inner membrane of mitochondria. The strongest bonds are formed by the sharing or transfer of electrons between atoms allowing the formation of molecules.
The electrode at which electrons are accepted by the oxidising agent is called the positive terminal. Ionic bonds are molecular unions formed when an electron-deficient atom a positive ion joins with an electron-excessive atom a negative ion. We saw this in the formation of NaCl.
Ubiquinone is called a mobile transfer molecule because it moves the electrons to the cytochrome b-c1 complex. In the simplest case the cation is a metal atom and the anion is a nonmetal atom but these ions can be of a more complicated nature eg. This transfer can be harnessed to form an electric current.
TRUE OR FALSE True. During the formation of some compounds atoms gain or lose electrons and form electrically charged particles called ions see figure below. In electron transfer the number of electrons lost must equal the number of electrons gained.