Transfer Reducing Equivalents

An electron transfer flavoprotein ETF or electron transfer flavoprotein complex CETF is a flavoprotein located on the matrix face of the inner mitochondrial membrane and functions as a specific electron acceptor for primary dehydrogenases transferring the electrons to terminal respiratory systems such as electron-transferring-flavoprotein dehydrogenase.
Transfer reducing equivalents. The electron transport chain ETC is a series of protein complexes that transfer electrons from electron donors to electron acceptors via redox reactions both reduction and oxidation occurring simultaneously and couples this electron transfer with the transfer of protons H ions across a membraneThe electron transport chain is built up of peptides enzymes and other molecules. For example the translocation of protons by cytochrome c oxidase is powered by reducing equivalents provided by reduced cytochrome c. The two systems may simultaneously or mutually operate in the transfer of reducing equivalents from the cytosol to inside the mitochondria.
Two proposed mechanisms are 1 inhibition of mitochondrial Complex 1 with consequent compromised ATP and AMP homeostasis or 2 inhibition of mitochondrial glycerophosphate dehydrogenase mGPDH and thereby attenuated transfer of reducing equivalents from the cytoplasm to mitochondria resulting in a raised lactatepyruvate ratio and redox-dependent. Mitochondrial uptake of InsP3R-released Ca2 is fundamentally required to provide optimal bioenergetics by providing sufficient reducing equivalents to support oxidative phosphorylation. The transfer of analytical procedures in liquid chromatography LC is a.
Absence of this Ca2 transfer results in enhanced phosphorylation of pyruvate dehydrogenase and activation of AMPK which activates prosurvival macroautophagy. They can be. Transfer of reducing equivalents into mitochondria by the interconversions of proline and delta 1-pyrroline-5-carboxylate.
In yeast 2 related COX assembly genes SCO1 and SCO2 synthesis of cytochrome c oxidase enable subunits 1 and 2 to be incorporated into the holoprotein. β-oxidation in the peroxisome requires the use of a peroxisomal carnitine acyltransferase instead of carnitine acyltransferase I and II used by the mitochondria for transport of the activated acyl group into the mitochondria for further. Mammalian cytochrome c oxidase COX catalyzes the transfer of reducing equivalents from cytochrome c to molecular oxygen and pumps protons across the inner mitochondrial membrane.
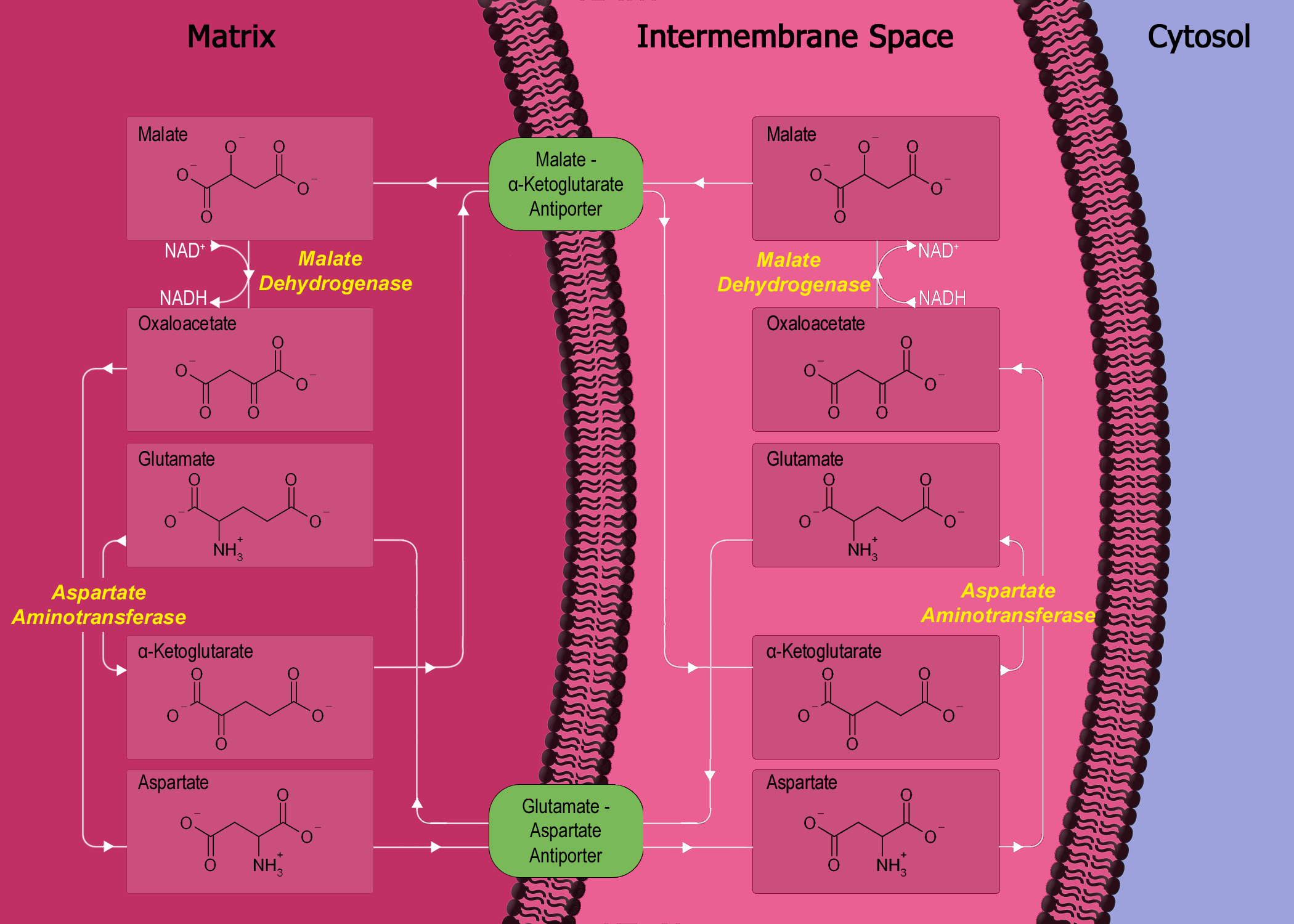
The uncoupling of intact cells by 24-dinitrophenol inhibits the translocation of reducing equivalents into the mitochondrial matrix probably by impairing the malate-aspartate shuttle. The use of mulches especially when the crop is small is another way of substantially reducing soil evaporation. A malate valve controls the export of reducing equivalents from the chloroplasts Chloroplasts are also able to export reducing equivalents by a malate-oxaloacetate shuttle via a specific malate-oxaloacetate translocator operating in a counter-exchange mode and located in the chloroplast inner envelope membrane.
Is simply obtaining equivalent results between both. For scenario B the method robustness is the focus since the method is transferred between two identical systems. In mitochondria reducing equivalents provided by electron transfer or photosynthesis power this translocation of protons.