Transferred Between A Conjugate Acid Base Pair

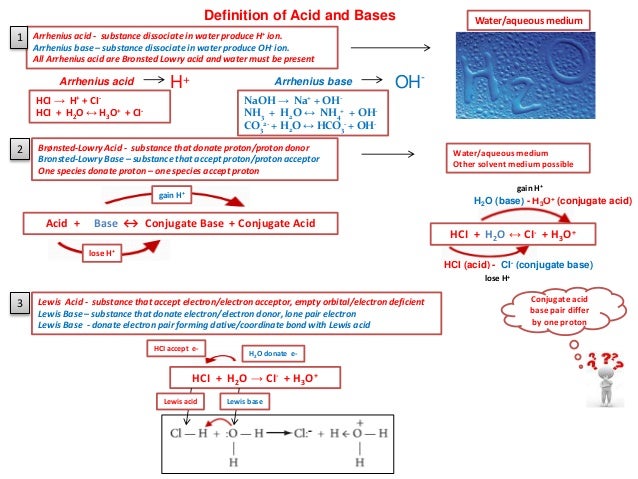
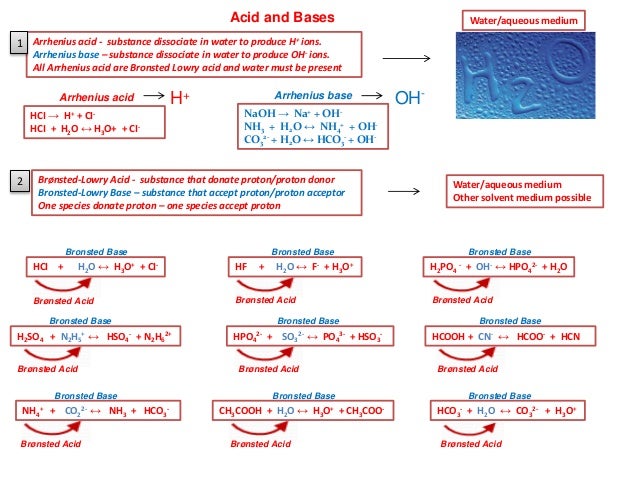
Every time a Bronsted acid acts as an H-ion donor it forms a conjugate base.
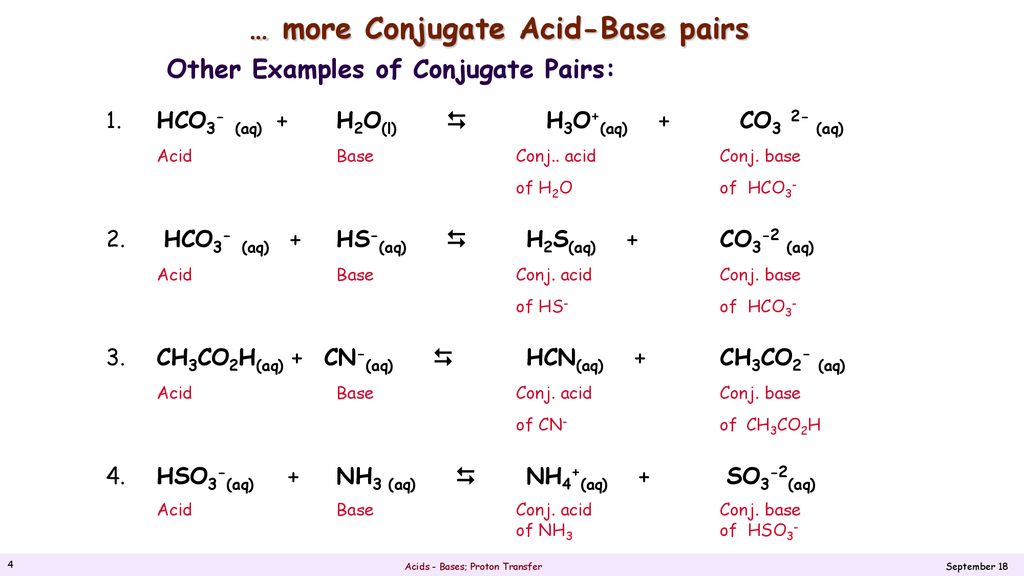
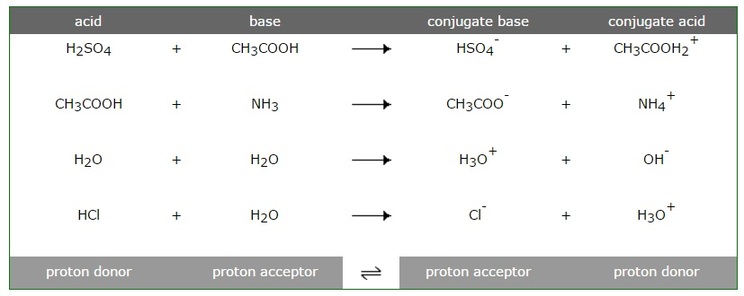
Transferred between a conjugate acid base pair. Conjugate Acid-Base Pairs One definition of acids and bases according to the Bronsted-Lowry theory involves an acid-base pair or conjugate pair. A conjugate acid within the BrnstedLowry acidbase theory is a chemical compound formed when an acid donates a proton H to a base in other words it is a base with a hydrogen ion added to it as in the reverse reaction it loses a hydrogen ion. 5192008 What is transferred between a conjugate acid-base pair.
An acid and a base which differ only by the presence or absence of a proton are called a conjugate acid-base pair. 832018 That is a conjugate acid and its conjugate base are related by the loss and gain of a hydrogen ion. The term conjugate comes from the Latin stems meaning joined together.
Imagine a generic acid HA. NH 4 NH 3. Sulfuric acid H 2 SO 4 has the sulfate ion SO 42- as the conjugate base.
Thus an acid-base reaction occurs when a proton is transferred from an acid to a base with formation of the conjugate base of the reactant acid and formation of the conjugate. Similarly HF is the conjugate acid of F and F the conjugate base of HF. And refers to things that are joined particularly in pairs such as Brnsted acids and bases.
How joyous movie stole Sundance broke sales record. By definition a conjugate acid-base pair is a pair of chemicals on either side of an acid-base equilibrium that is linked through a proton transfer. As an ion chloride becomes the conjugate base of HCl and hydronium is the conjugate acid of H 2 O.
Chemists sometimes call a hydrogen ion a proton. Given the reaction above which compound is the bronsted-Lowry Conjugate base if HCl is the acid. The dissociation of HCl in water.