Bonding By Transfer Of Electrons Guide Questions
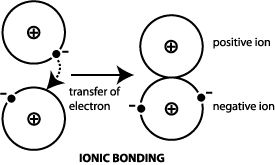
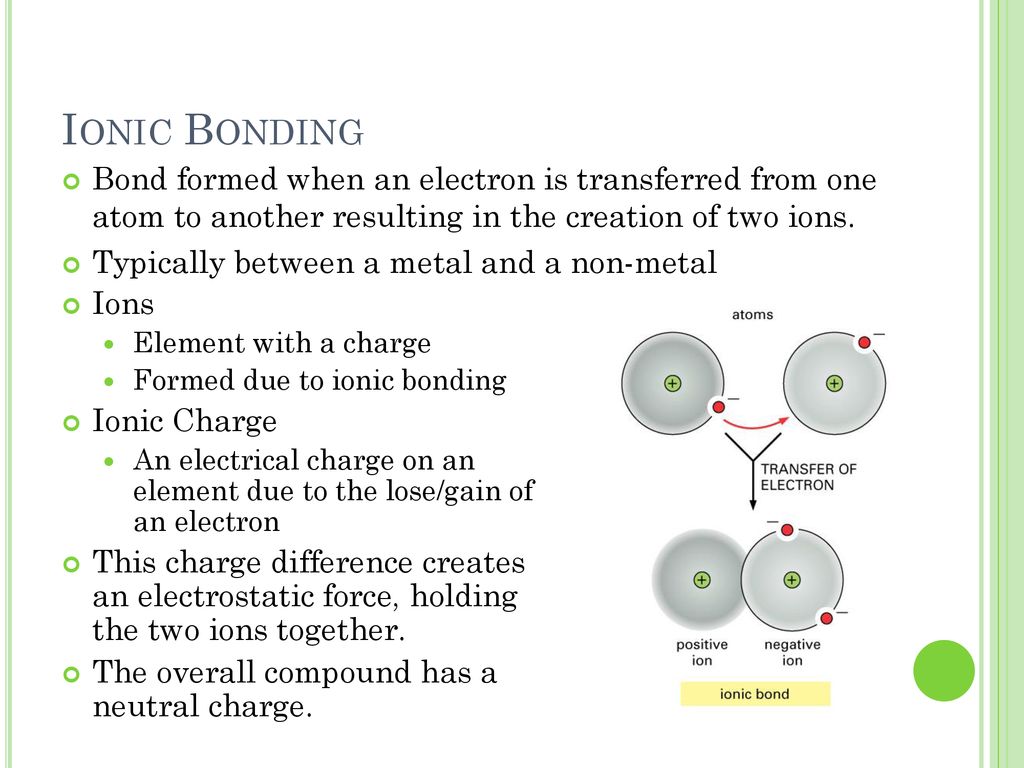
The ionic bond involves a transfer of electrons.
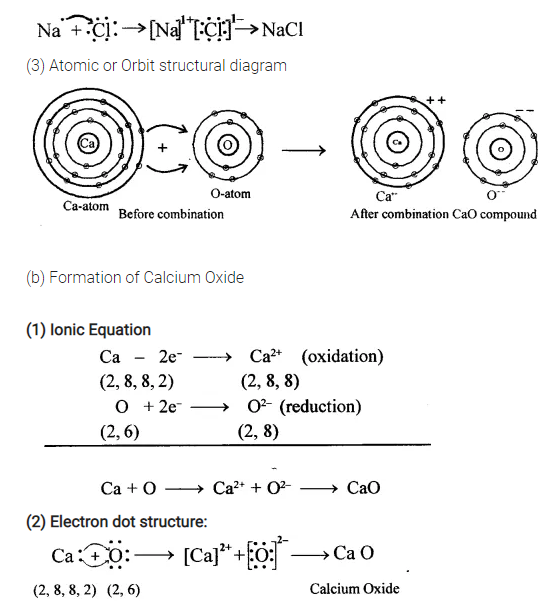
Bonding by transfer of electrons guide questions. Formed when one or more electrons are transferred from one ato. The attraction between these two oppositely charged ions is known as ionic bond. Mg goes from 1s2 2s2 2p63s2 to Mg2 1s2 2s2 2p6 O goes from 1s2 2s2 2p4 to O2-1s2 2s2 2p6.
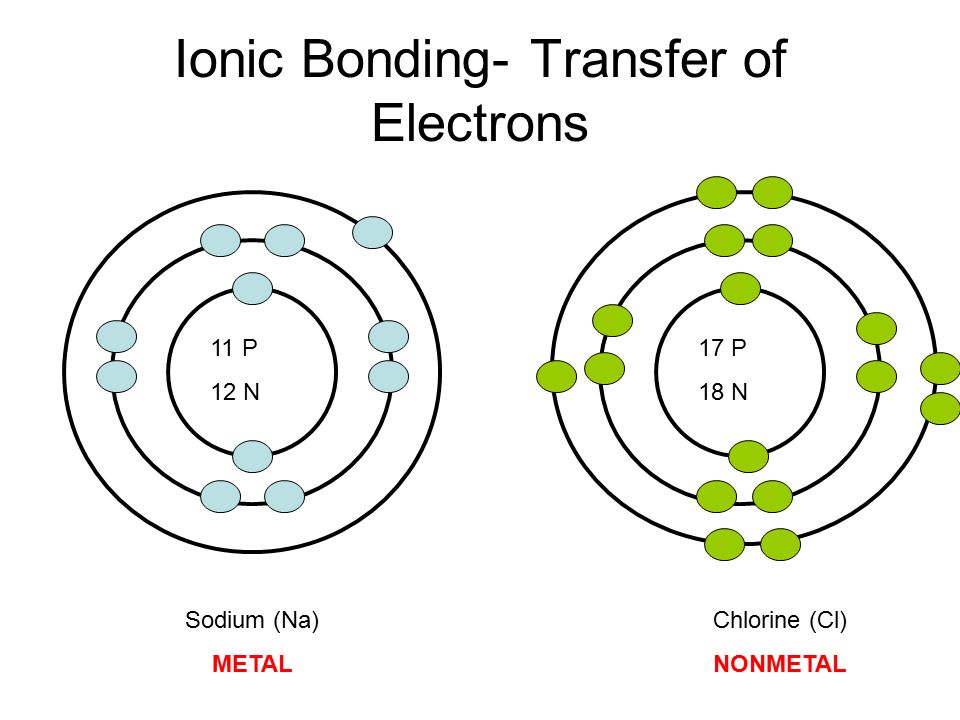
Metal atoms lose electrons to form ve ions. Learn test study guide chemistry bonding with free interactive flashcards. 1302021 Ionic bonding involves a complete transfer of an electron from one atom to another so orbital overlap or hybrization does not occur.
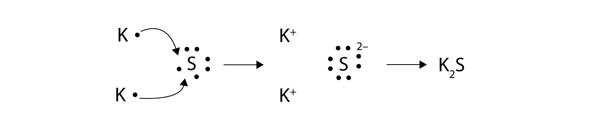
The chemical formula for magnesium fluoride is MgF 2 reflecting what has happened in the bonding. The metal atom loses one or more electrons to form a positive ion cation. A similar process occurs between Mg atoms and O atoms except in this case two electrons are transferred.
In ionic bonding which involves electrostatic forces between atoms of opposite charge or differing electronegativity rather than the sharing of electrons one atom donates an electron and another receives one. Molecules having uneven distribution of electrons and charges. A strong force of attraction is holding atoms together in a mo.
The transfer of electrons. Ionic bond - a bond that occurs between atoms through the transfer of electrons when a positively charged atom and negatively charged atom. Covalent bonding in which neither atom loses complete control over its valence electrons is.
For instance sodium chloride is formed when. Ion - an atom with a positive or negative charge. Mg goes from 1s2 2s2 2p63s2 to Mg2 1s2 2s2 2p6 O goes from 1s2 2s2 2p4 to O2-1s2 2s2 2p6 Definition.