The Transfer Or Sharing Of Electrons Is Called

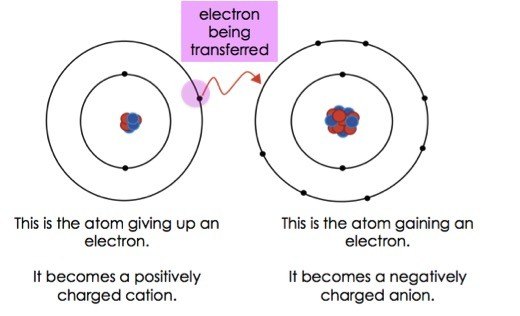
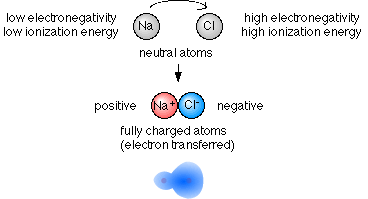
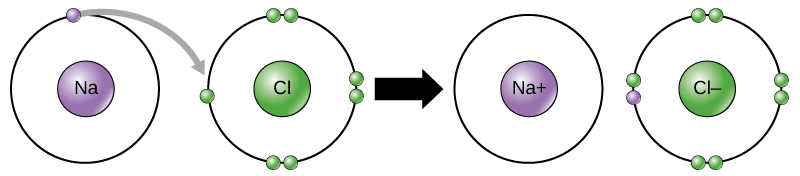
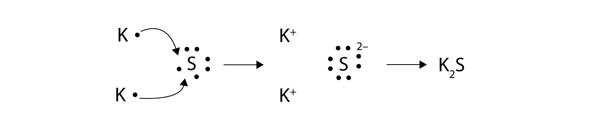
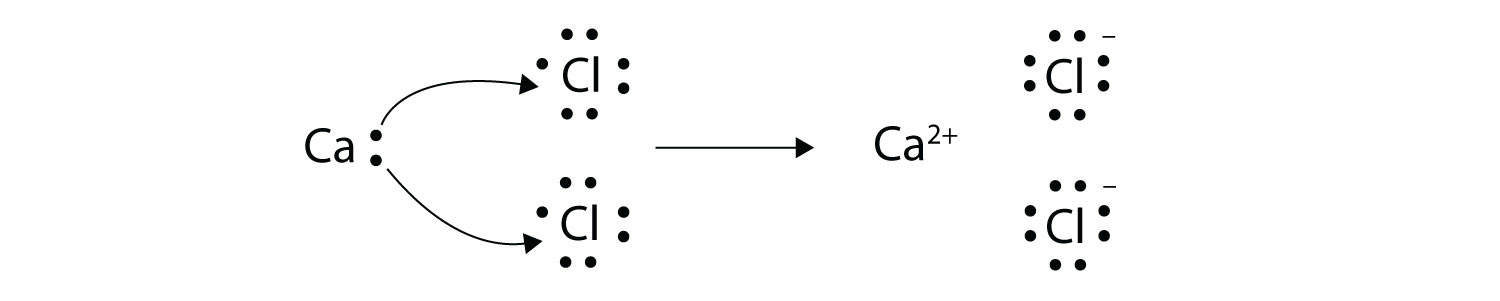
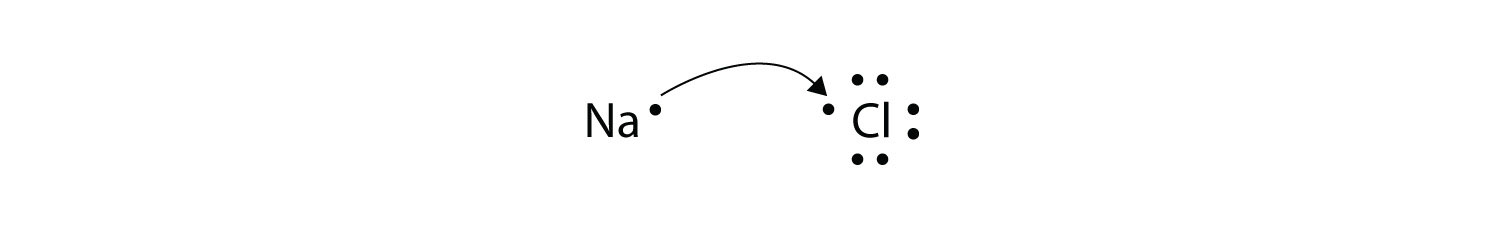
An ionic bond is also called an electron covalent bond formed by the complete transfer of some electrons from the outermost orbital from one atom to another.
The transfer or sharing of electrons is called. The Chemistry of Life. Choice c is. How single bonds are formed.
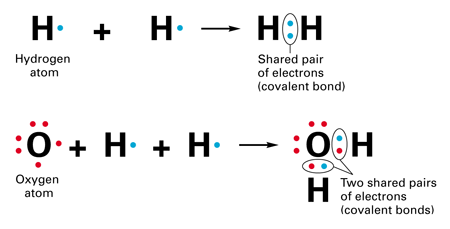
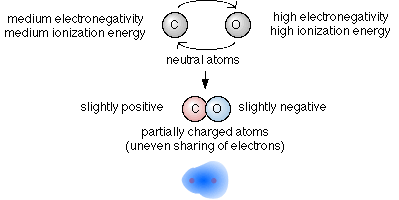
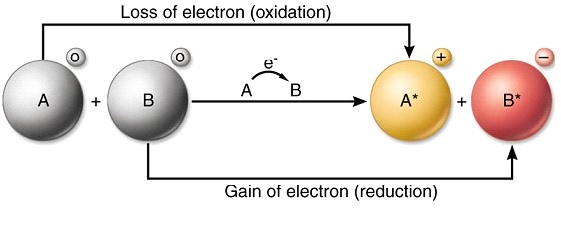
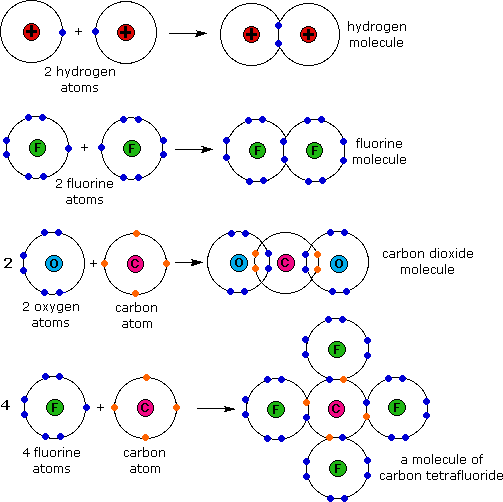
In covalent bonding the electrons are shared to varying degrees between atoms. Chemical bonds formed by the sharing of electrons are called covalent bonds. - Electrons are transferred or shared in such a way that each atom acquires an especially stable electron configuration.
A A sodium atom Na has equal numbers of protons and electrons 11 and is uncharged. Specifically a shared-electron bond is called a covalent bond. Sodium atoms in Group 1A 1 are neutral with 11 electrons and 11 protons.
The compound which is formed after sharing of a pair of electrons is called Covalent bonds. The bond involving transfer of electrons is ionic. Electrons involved with bonding move toward the atom with the higher _____.
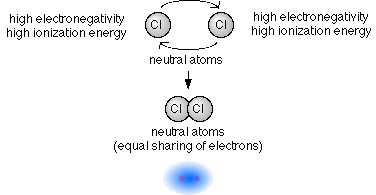
Ionic _____ bonds involve the sharing of electrons between atoms. A bond formed by the sharing of electrons between atoms is called a covalent bond. Electron transfer would be more like ionic bonding where two.
2Covalent bond- Some electrons from each atom are shared by all the atoms such that those electrons complete the octet of each contributing atom. Usually this is a noble gas configuration one with eight valence outer-shell electrons or an octet. A 3D object consisting of repeating patterns of alternating positive and negative ions.