The Transfer Or Sharing Of Valence Electrons Creates

Some key characteristics of a valence electron are.
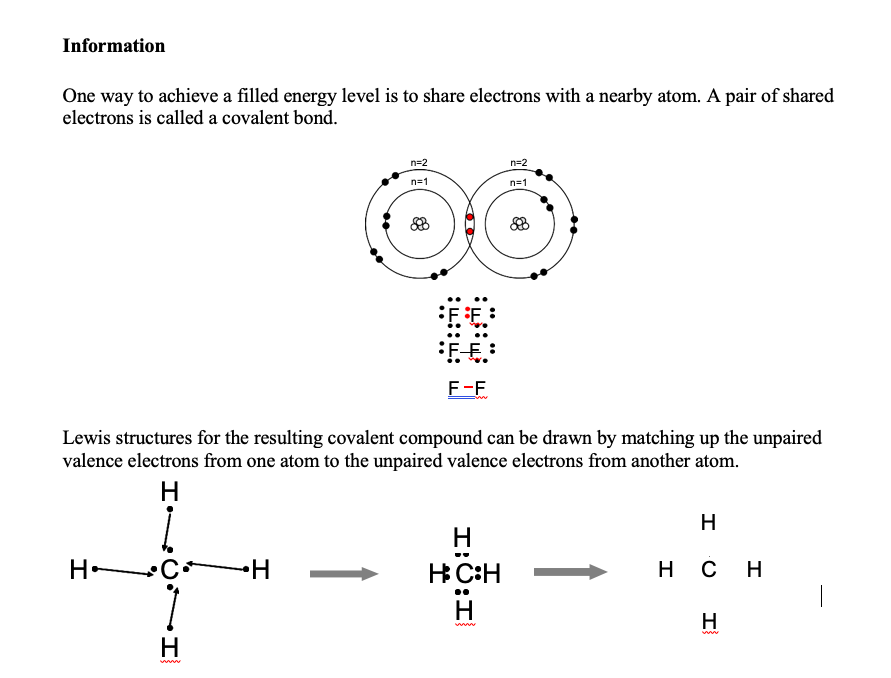
The transfer or sharing of valence electrons creates. The slight influence of an external force can easily detach the electron from its orbit. 6202014 Ionic bonding is the complete transfer of valence electron s between atoms. 1232021 Realize that valence electrons can be represented by dots.
What is the number of valence electrons for Oxygen. Electrons however can be added to atoms by transfer from other atoms lost by transfer to other atoms or shared with other atoms. Be sure to draw extra atoms if one of the atoms cant accept anymore.
The transfer of electrons results in what is known as an ionic bond between atoms while sharing of electrons creates. You just studied 7 terms. Which of the following statements is true about covalent bonds.
Other articles where Electron transfer is discussed. Determine which electrons will need to share to create a pair and a full octet on each element. Know that covalent bonding involves two nonmetals or a metalloid with a nonmetal sharing electrons.
Which type of bond creates a pool. However it is a different sort of bonding than covalent bonding. Form a silicon crystal.
A silicon Si atom with its four valence electrons shares an electron with each of its four neighbors. Know that there are two types of bonding. For many molecules the sharing of electrons allows each atom to attain the equivalent.