Transfer Of Electrons Electronegativity

This attraction is known as an ionic bond.
Transfer of electrons electronegativity. The rule is that when the electronegativity difference is greater than 20 the bond is considered ionic. Substances such as NaCl and MgCl 2 are the usual examples. At the macroscopic scale ionic compounds form lattices are crystalline solids under normal conditions and have high melting points.
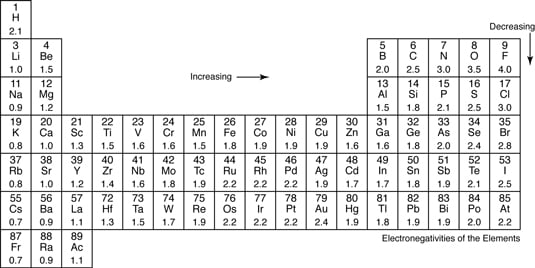
An Introduction to General Organic and Biological Chemistry 12th Edition answers to Chapter 6 - Section 61 - Ions. The most electronegative atom fluorine is assigned a value of 40 and values range down to cesium and francium which are the least electronegative at 07. The higher the associated electronegativity number the more an element or compound attracts electrons towards it.
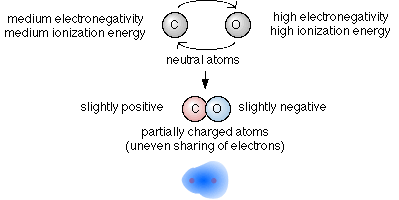
But if the electronegativity difference between the bonding atoms is too large it results in a complete transfer of electrons. The transfer of electrons between carbon C and oxygen O allows the storage and release of energy transmitted to Earth from the Sun. The greater the difference between atom electronegativity values the more polar the chemical bond formed between them.
Electronegativity is the tendency of an atom to attract electrons in a molecule. 1242020 Electronegativity is an atoms tendency to attract electrons to itself in a chemical bond. The two idealized extremes of chemical bonding.
Ionic Versus Covalent Bonding Ionic compoundsare formed when electrons are transferredfrom one atom to another The transfer of electrons forms ions Each ion is isoelectronic with a noble gas Electrostatic force ionic bond holds atoms together Covalent bondinginvolves sharingof electrons to achieve noble gas configurations for the atoms involved. Ionic bonds are formed by the transfer of electrons between a non metal and a metal. 61b Answer Calcium must lose two electrons to achieve noble gas configuration.
First Ionization Energy of Cobalt. We must keep the phenomenon in the bulk of the solution separate from the phenomenon at the electrodes. Electronegativity is a measure of the attraction of a bonded atom for the pair of electrons in a covalent bond.