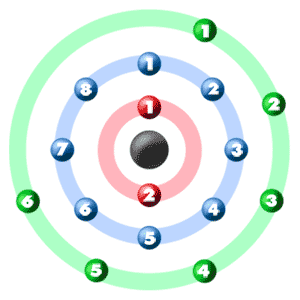
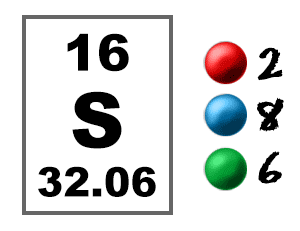
Transfer Of Electrons Sulfur

With oxidation numbers inserted as superscripts this reaction is written.
Transfer of electrons sulfur. Then draw arrows to show the transfer of electrons. NADH dehydrogenase Transfer of Electrons from NADH to Coenzyme Q It is the first complex of the electron transport chain. Ubiquinone Co-enzyme-Q Between the flavoproteins and cytochromes are other electron carriers termed ubiquinone UQ.
In the cytosolic ironsulfur protein assembly machinery two human key proteinsNADPH-dependent diflavin oxidoreductase 1 Ndor1 and anamorsinform a stable complex in vivo that was proposed to provide electrons for assembling cytosolic ironsulfur. IONIC BONDING Answer Key Points. The sulfate absorbed by roots from soil solution is translocated through xylem to shoots for assimilation.
However a few bacterial ferredoxins of the 2 4Fe4S type have two iron sulfur clusters and can carry out two electron transfer reactions. The electrostatic force of attraction will then bring the magnesium cations and the sulfur anions together an ionic bond is formed. Depending on the sequence of the protein the two transfers can have nearly identical reduction potentials or they may be significantly different.
TRUE Sulfur possesses a δ and oxygen possesses a δ. During this process of transfer of an electron the iron in heme. To show that both elements change oxidation numbers.
The electrons are further transferred to a series of iron-sulfur complexes Fe-S which have a higher relative affinity towards the electrons. C Name the ions present in this compound CaS. FALSE The electrons are equally shared between sulfur and oxygen.
Sulfur oxidation to sulfate coupled with electron transfer to electrodes by Desulfuromonas strain TZ1. 1112020 In high energy density LiS battery the effective anchoring of sulfurLiPS and the timely transfer of Li-ionselectrons are necessary to ensure the high sulfur utilization in the electrochemical reaction especially in the thick and dense electrode with lean electrolyte. Because the oxidation numbers changed an oxidationreduction reaction is defined as one in which electrons are transferred between atoms.