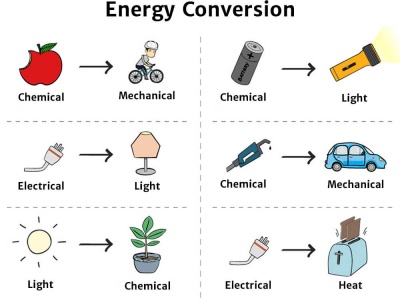
Transfer Of Energy From One System To Another

392021 Energy is the ability of a system to do work.
Transfer of energy from one system to another. Different forms of energy such as heat light and motion can be transmitted. Thermodynamically heat energy is defined as the energy transferred from one system to another that is not work. Thermodynamically heat energy is defined as the energy transferred from one system to another that is not doing work.
Energy transfer takes place when energy moves from one place to another. 852015 The transfer of energy from one object to another occurs when there is a reaction such as during a collision according to the Chemical Education Digital Library. First chemical energy stored in coal or fossil fuels is released by burning it.
Like electricity traveling through a superconductor not all interactions actually result in an energy transformation. The chemical energy is converted to heat energy and light energy. A system can transfer energy to another system by simply transferring matter to it since matter is equivalent to energy in accordance with its mass.
Energy transfer is the movement of energy from one location to another. Some thermal energy in all materials is transformed into light energy and radiated into the environment by electromagnetic waves. While energy can be transferred or transformed the total energy always remains the same.
The answer is from a series of energy conversions. Energy is transferred from one object to another when a reaction takes place. The transfer of energy from one body region or thermodynamic system to another thermal energy the total internal energy of a thermodynamic system or substance that is measured as the systems temperature.
Thermal energy can be transferred by conduction convection and radiation. Energy from the chemical store in the gas increases the internal thermal energy of the water in the pan. 842015 Energy changes from one form to another by interacting with objects in its environment that resist the influence of that energy.