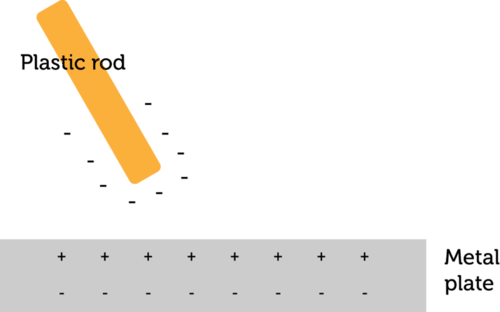Transfer Of Electrons Materials
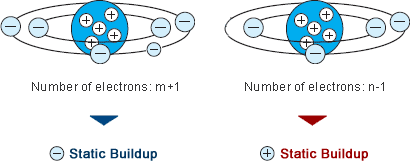
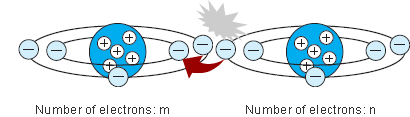
Rubbing two surfaces together increases the transfer of electrons because it creates a closer contact between the materials.
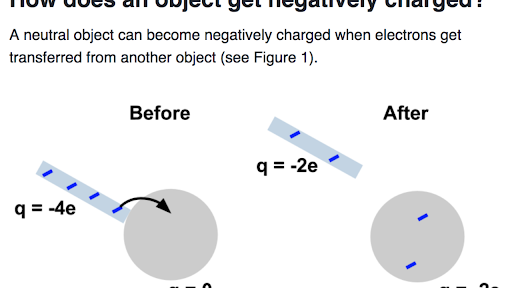
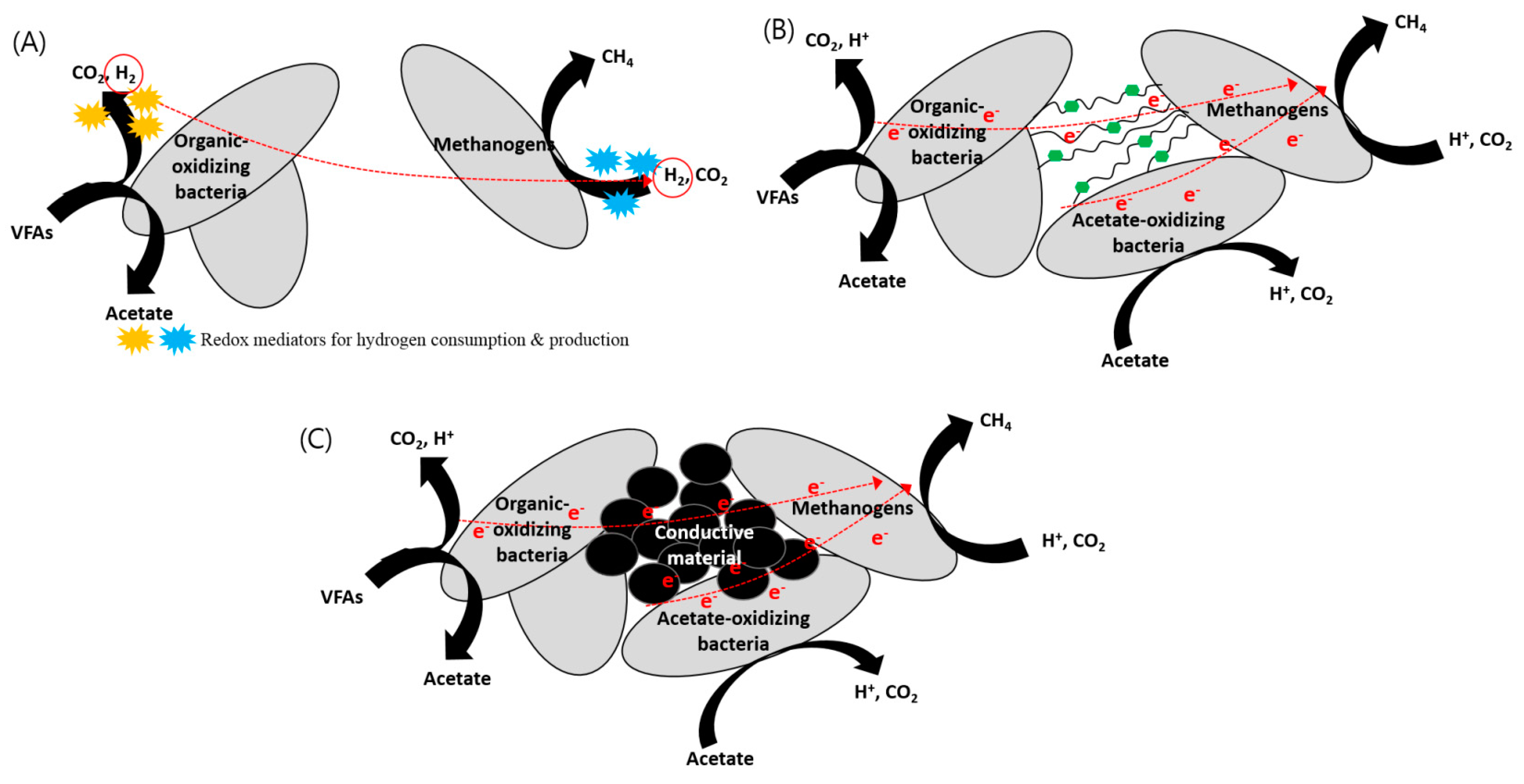
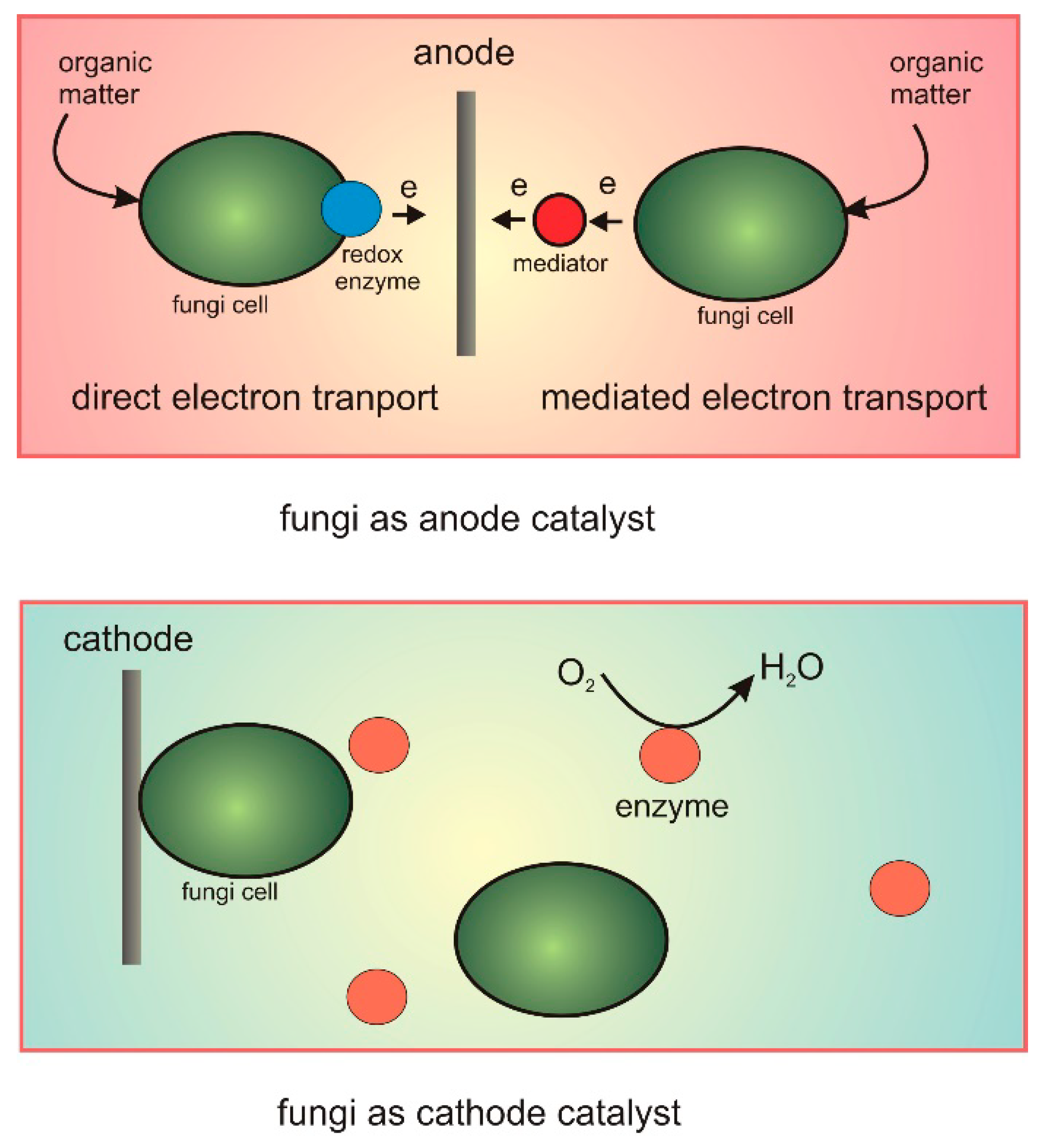
Transfer of electrons materials. Thus when you walk across a carpet on a dry day your shoes rub against the carpet and some electrons are removed from the carpet by your shoes. While external forces such as physical rubbing can force some of these electrons to leave their respective atoms and transfer to the atoms of another material they do not move between atoms within that material very easily. Electron transfer take place when bacteria are in contact with electrically conductive materials and ROS is believed to be controlled and consumed sequentially in the electron transfer process.
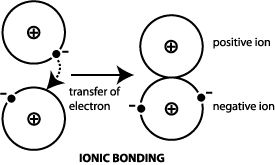
MIT researchers are working to develop an electron-cloaking material that would hide particles from passing electrons possibly leading to more efficient thermoelectric devices and new. Electrons emitted in this manner are called photoelectrons. A sodium atom can assume the electron structure of neon by a transfer of its one valence 3s electron to a chlorine atom.
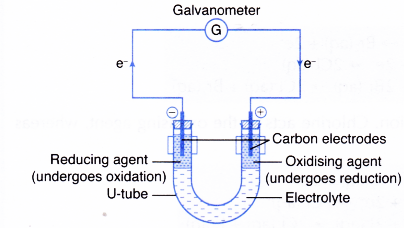
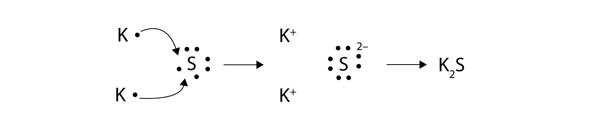
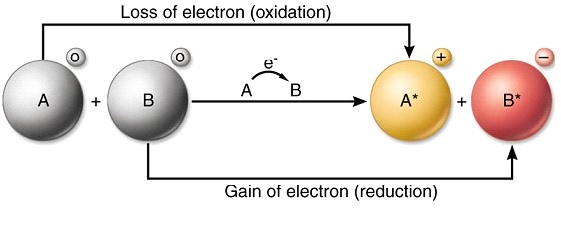
To investigate oxidation and reduction in the transfer of electrons at a distance. 9162014 With arrows illustrate the transfer of electrons to form calcium chloride from Ca atoms and Cl atoms. The effect has found use in electronic.
Normally electrons travel through a material in a way that is similar to the motion of electromagnetic waves including light. Nanobiomaterials in Clinical Dentistry Second Edition 2019. The physical nature of potential and kinetic energy in solids as well as their enthalpy entropy and Helmholtz free energy was previously considered.
The phenomenon is studied in condensed matter physics and solid state and quantum chemistry to draw inferences about the properties of atoms molecules and solids. 20 mol dm -3 sulphuric acid 05 mol dm -3 freshly prepared ironII sulphate solution 02 mol dm -3 acidified potassium manganateVII solution 05 mol dm -3 potassium iodide solution 02 mol dm -3 acidified potassium dichromateVI solution 02 mol dm -3 potassium. The transfer of energy in the form of electrons allows the cell to transfer and use energy in an incremental fashionin small.
Plasmonic photocatalysis on bimetallic heterostructures via plasmonic hot electrons eph has received significant attention owing to their ability to harvest solar energy and high catalytic activities whereas the mechanism of the eph transfer at the metalmetal interfaces is still not completely understo Journal of Materials Chemistry C HOT Papers. After such a transfer the chlorine ion acquires a net negative charge an electron configuration identical to that of argon. That led the MIT researchers to the idea of harnessing the cloaking mechanisms developed to shield objects from view but applying it to the movement of electrons which is key to electronic and.